Advertisements
Advertisements
Question
(a) Calculate the heat capacity of a copper vessel of mass 150g if the specific heat capacity of copper is `410 J kg^-1 K^-1`
(b) How much heat energy will be required to increase the temperature of the vessel in part (a) from 25°C to 35°C?
Solution
Mass of copper vessel = 150 g
= 0.15 kg
The specific heat capacity of copper = `410 J kg^-1K^-1`.
Heat capacity = Mass × specific heat capacity
= `0.15 kg × 410Jkg^-1K^-1`
= `61.5JK^-1`
Change in temperature= (35 − 25)°C = 10°C = 10K
(ii) Energy required to increase the temperature of vessel
ΔQ = mcΔT
= 0.15 × 410 × 10
= 615 J
APPEARS IN
RELATED QUESTIONS
What is the other name given to the principle of the mixtures?
Name the law on which the principle of mixtures is based
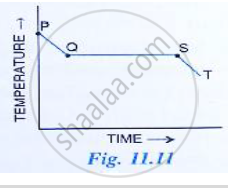
Fig 11. 11 shows the variation in temperature with time when some wax cools from the liquid phase to the solid phase.
(i) In which part of the curve, the wax is in liquid phase?
(ii) What does the part QS of the curve represent?
(iii) In which part of the curve, the wax will be the in the liquid as well as solid phase?
(iv) In which part of the curve, the wax is in solid phase?
A bucket contains 10 liters of water at 80°C. Cold water at 25°C is run from a tap into the bucket of hot water for 20 seconds and the temp. Of the water in the bucket falls to 50°C.
(a) Calculate the rate at which cold water came out of the tap.
(b) State an assumption made in the above calculation.
Ice is more effective in cooling than the ice-water. Explain.
Why does atmospheric temperature fall after hail storm?
A copper calorimeter of mass 50g contains 100g of water at 20°C. A metallic piece of mass 250 g is heated to 100°C and is then dropped into the calorimeter. The contents of the calorimeter are well stirred and its final highest temperature is recorded to be 28 °C. If the specific heat capacity of water is 4.2 J/g°C and of copper is 0.4 J/g°C, find:
(i) the heat gained by water,
(ii) the heat gained by calorimeter,
(iii) total heat supplied by the metal piece, and
(iv) the specific heat capacity of metal.
A calorimeter is a device used to measure the ______.
