Advertisements
Advertisements
Question
Choose the correct words to fill in the blanks.
Cations are formed by _______ (loss/ gain) of electrons and anions are formed by ________( loss/gain) of electrons.
Solution
Cations are formed by loss of electrons and anions are formed by gain of electrons.
APPEARS IN
RELATED QUESTIONS
Identify the substance underlined, in the following case
The particles present in a liquid such as kerosene, that is a nonelectrolyte.
Define the following term:
Non-electrolyte
Explain the term : Electro metallurgy
Fill in the blank.
As we descend the electrochemical series containing cations, the tendency of the cations to get ________ at the cathode increases.
A solution contains magnesium ions (Mg+2), iron (II) ions (Fe+2) and copper ions (Cu+2). On passing an electric current through this solution, which ions will be the first to be discharged at the cathode? Write the equation for the cathode reaction.
Why is carbon tetrachloride, which is a liquid, a non-electrolyte?
A metal article is to be electroplated with silver. The electrolyte selected is sodium argentocyanide.
- What kind of salt is sodium argentocyanide?
- Why is it preferred to silver nitrate as an electrolyte?
- State one condition to ensure that the deposit is smooth, firm and long lasting.
- Write the reaction taking place at the cathode.
- Write the reaction taking place at the anode.
Select the correct answer from the choices a,b,c and d which are given. Write only the letter corresponding to the correct answer.
A compound which liberates reddish brown gas around the anode during the electrolysis in its molten state is:______________
Copper sulphate solution is electrolyzed using copper electrodes. Study the diagram given alongside and answer the questions that follow.
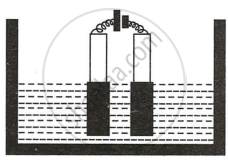
- Which electrode to your left or right is known as the oxidizing electrode and why?
- Write the equation representing the reaction that occurs.
- State two appropriate observations for the above electrolysis reactions.
What is the difference between:
Strong electrolyte and weak electrolyte.
