Advertisements
Advertisements
Question
A metal article is to be electroplated with silver. The electrolyte selected is sodium argentocyanide.
- What kind of salt is sodium argentocyanide?
- Why is it preferred to silver nitrate as an electrolyte?
- State one condition to ensure that the deposit is smooth, firm and long lasting.
- Write the reaction taking place at the cathode.
- Write the reaction taking place at the anode.
Solution
- It is a complex salt.
- Sodium argentocyanide is preferred over silver nitrate because it gives a smooth and firm silver plating over the article.
- If silver nitrate is used, then the deposition of silver will be fast and not smooth.
- Reaction at Cathode:
\[\ce{Ag+ + e- -> Ag}\] - Reaction at Anode:
\[\ce{Ag − e- -> Ag+}\]
APPEARS IN
RELATED QUESTIONS
Name a non-metallic element which is a conductor of electricity.
Define the term : Electrolyte
Write the difference between with examples:
Electrolytic dissociation and ionization
Make a neatly labeled sketch to show how a brass spoon can be plated with silver. Explain your choice of electrolyte used.
How is the passage of electricity through an electrolyte different from the passage of electricity through a copper wire?
Explain, why solid sodium chloride does not allow electricity to pass through?
Copper sulphate solution is electrolyzed using copper electrodes. Study the diagram given alongside and answer the questions that follow.
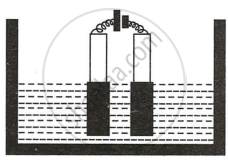
- Which electrode to your left or right is known as the oxidizing electrode and why?
- Write the equation representing the reaction that occurs.
- State two appropriate observations for the above electrolysis reactions.
State the observation at the anode and at the cathode during the electrolysis of :
Fused lead bromide using graphite electrodes
If HX is a weak acid, what particles will be present in its dilute solution apart from those of water?
Copper sulphate solution is electrolysed using copper electrodes.
Write the equation for the reaction occurring at the Anode electrode.
