Advertisements
Advertisements
Question
Choose the correct option from given alternative:
When sodium hydrogen carbonate is added to ethanoic acid a gas evolves. Consider the following statements about the gas evolved?
(A) It turns lime water milky.
(B) It is evolved with brisk effervescence.
(C) It has a smell of burning sulfur.
(D) It is also a by-product of respiration.
Options
(A) and (B) only
(B) and (D) only
(A), (C) and (D)
(A), (B) and (D)
Solution
When sodium carbonate is reacted with ethanoic acid it forms sodium ethanoate, water, and carbon dioxide gas. Carbon dioxide can be identified by the brisk effervescence. It turns lime water milky and is also a byproduct of respiration.
\[\ce{CH3COOH + NaHCO3 -> CH3COONa + H2O + CO2}\]
Hence, the correct answer is an option (A), (B) and (D).
APPEARS IN
RELATED QUESTIONS
What do you notice when acetic acid reacts with litmus?
What do you notice when acetic acid reacts with alkalies?
Fill in the blank with appropriate word/words.
Vinegar is a solution of about ________per cent ________in water.
Draw the structural formula of a compound with two carbon atoms in the following case:
An alcohol containing two carbon atoms.
State how the following conversions can be carried out:
Ethyl chloride to Ethene.
Give balanced chemical equations for the following conversion :
Ethanoic acid to ethyl ethanote
Write the characteristics of ethanoic acid.
Give the balanced chemical equation of the following reaction:
Neutralization of NaOH with ethanoic acid.
In the presence of the acid catalyst, ethanoic acid reacts with ethanol and ______ ester is produced.
Observe the diagram given below and answer the questions:
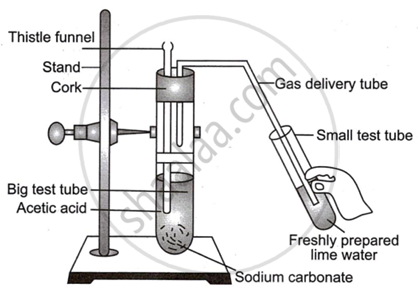
- Name the reactants in this reaction.
- Which gas comes out as effervescence in the bigger test tube?
- What is the colour change in the lime water?
- In the above experiment instead of sodium carbonate which chemical can be used to get same products?
- Write the use of acetic acid.
