Advertisements
Advertisements
Question
Complete the following flowchart.
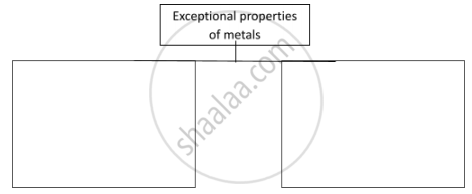
Solution
| Exceptional properties of metals | |
| Properties | Exceptions |
|
1. Physical state: Metals exist mainly in a solid state at room temperature. 2. Hardness: Metals are generally hard. 3. Melting and boiling point: Metals have high melting and boiling points. |
1. Mercury and gallium exist in a liquid state at room temperature. 2. Lithium, sodium and potassium are metals which can be cut with a knife as they are very soft. 3. Sodium, potassium, mercury, and gallium have very low melting and boiling points. |
APPEARS IN
RELATED QUESTIONS
Mark ‘T’ if the statement is true and ‘F’ if it is false.
Generally, non-metals react with acids. ( )
Saloni took a piece of burning charcoal and collected the gas evolved in a test tube.
(a) How will she find the nature of the gas?
(b) Write down word equations of all the reactions taking place in this process.
Which property of graphite is utilised in making electrodes?
State one use of Gold.
Explain how, a reduction reaction of aluminium can be used for welding cracked machine parts of iron. Write a chemical equation for the reaction involved.
What is an alloy? How is an alloy made?
How are the properties of an alloy different from those of the constitutent elements?
Give two uses of Germanium
Fill in the blank spaces by choosing the correct words from the given list.
List: amalgam, chlorine, gallium, sodium chloride, silver chloride
______ is a liquid metal.
Why is hydrogen placed with alkali metals?
What is it made from? Why?
Pots and pans
With reference to the following metals state their use in daily life metal:
copper
Give a reason why iron objects rust easily but the iron pillar at Qutab Minar has not rusted over the years.
The metal which is liquid at room temperature is ______.
Compare the properties of metals and nonmetals. Give three examples for each.
Which of the following two combinations are correct?
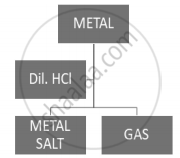
| Metal | Gas Evolved | |
| (i) | Copper | Yes |
| (ii) | Iron | Yes |
| (iii) | Magnesium | No |
| (iv) | Zinc | Yes |
Name one metal and one non-metal that exist in liquid state at room temperature. Also name two metals having melting point less than 310 K (37°C)
An element forms an oxide A2O3 which is acidic in nature. Identify A as a metal or non-metal.
In the given figure, you find that the bulb glows when an iron nail is placed between two ends of wire. Complete the following sentences on the basis of this fact.

- ______ is a metal.
- Metals are good ______ of electricity.
Compare the conductivity of metals and non–metals.
