Advertisements
Advertisements
Question
Define hydrogen bond. Is it weaker or stronger than the van der Waals forces?
Solution
A hydrogen bond is defined as an attractive force acting between the hydrogen attached to an electronegative atom of one molecule and an electronegative atom of a different molecule (may be of the same kind).
Due to a difference between electronegativities, the bond pair between hydrogen and the electronegative atom gets drifted far away from the hydrogen atom. As a result, a hydrogen atom becomes electropositive with respect to the other atom and acquires a positive charge.
`4^"g" - "X"^("g"-)......."H"^("g+") -"X"^("g-")......."H"^("g+") -"X"^("g-")`
The magnitude of H-bonding is maximum in the solid state and minimum in the gaseous state.
There are two types of H-bonds:
(i) Intermolecular H-bond e.g., HF, H2O etc.
(ii) Intramolecular H-bond e.g., o-nitrophenol
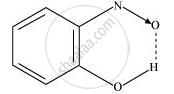
Hydrogen bonds are stronger than Van der Walls forces since hydrogen bonds are regarded as an extreme form of dipole-dipole interaction.
APPEARS IN
RELATED QUESTIONS
For decolourisation of 1 mole of acidified KMnO4, the moles of H2O2 required is
The type of H-bonding present in ortho nitrophenol and p-nitrophenol are respectively
Arrange NH3, H2O and HF in the order of increasing magnitude of hydrogen bonding and explain the basis for your arrangement.
In the following compounds, the decreasing order of basic strength will be ______.
