Advertisements
Advertisements
Question
Describe an experiment to prove the following:
HCI gas contains the element chlorine.
Solution
When HCI gas is heated above 500°C, it dissociates into hydrogen and chlorine.
\[\ce{2HCl ⇌[>500°C] H2 + Cl2}\]
APPEARS IN
RELATED QUESTIONS
Write a balanced chemical equation for the action of hydrochloric acid on sodium bicarbonate.
Name the acid used for the preparation of hydrogen chloride gas in the laboratory. Why is this particular acid preferred to other acids?
Write the balanced chemical equation for the laboratory preparation of hydrogen chloride gas.
Explain why thick white fumes are formed when a glass rod dipped in \[\ce{NH4OH}\] is brought near the mouth of a bottle full of \[\ce{HCl}\] gas.
Explain why dry hydrogen chloride gas does not affect a dry strip of blue litmus paper but it turns red in the presence of a drop of water.
The given set up in the figure is for the preparation of an acid.
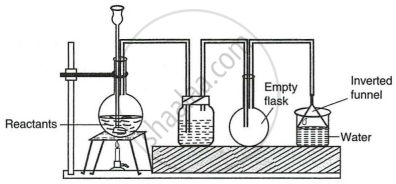
- Name the acid prepared by this method.
- Name the reactants used.
- Why an empty flask is used?
- What is the drying agent used? Why is this drying agent chosen?
- What is the role of the inverted funnel in the arrangement?
Name the gas produced when chlorine water is exposed to sunlight.
The drying agent used to dry \[\ce{HCl}\] gas is ______.
What are the important precautions?
Explain, why (or give reasons for)
Hydrogen chloride is not collected over water.
Mention the reaction condition and give balanced equation to obtain: Cl2 gas from HCI acid.
Mention the reaction condition and give balanced equation to obtain : HCl gas from common salt
Choose the correct answer from the options given below:
Dilute hydrochloric acid solution cannot be concentrated by boiling beyond
Complete and balance the equations:
KMnO4 + HCI → _______+ _______ + _______ + ______ + 8H2O
Write balanced equation for the reaction between dilute hydrochloric acid and sodium sulphite.
One chemical test that would enable you to distinguish between the following pair of chemicals. Describe what happens with each chemical or state 'no visible reaction'.
Sodium chloride solution and sodium nitrate solution.
Answer the following question related to the laboratory preparation of the hydrogen chloride gas:
Why is sodium chloride preferred to other metallic chlorides?
Answer the following question related to the laboratory preparation of the hydrogen chloride gas:
Write the chemical equation.
Complete and balance the following reaction, state whether dilute or conc. acid is used.
\[\ce{NH4OH + HCl ->}\]
Identify the gas evolved in the following reaction:
\[\ce{MnO2}\] reacts with concentrated \[\ce{HCl}\].
