Advertisements
Advertisements
Question
Differentiate between:
Aqueous ferrous and ferric sulphate solution
Solution
| Aqueous ferrous | Ferric sulphate solution |
|
When ammonium solution is added to aqueous solution of ferrous sulphate a dirty green precipi tate of ferrous hydroxide is obtained which is insoluble in excess of ammonium solution. Hydroxide. \[\ce{FeSO4 + 2NH4OH -> (NH4)2SO4 + Fe(OH)2}\] |
On adding ammonium solution to ferric sulphate solution we get ammonium sulphate and reddish brown precipi tate of ferric hydroxide which is insoluble in excess of ammonium solution . \[\ce{Fe2(SO4)3 + 6NH4OH -> 3(NH4)2SO4 + 2Fe(OH)3}\] |
APPEARS IN
RELATED QUESTIONS
Is ammonia more dense or less dense than air?
Mention two possible ways by which ammonia produced is removed from unchanged gases.
Describe two tests to identify ammonia and ammonium ions in an aqueous solution.
The diagram shows a simple arrangement of the fountain experiment:
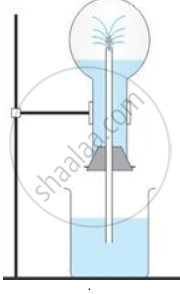
(i) Name the two gases you have studied which can be used in this experiment
(ii) What is the common properly demonstrated by this experiment
State one relevant observation of burning of ammonia in air.
What do you observe when Ammonium solution is added to ferric chloride solution.
Choose the correct answer from the options given below :
Ammonia is produced when ammonium chloride is heated with
Of the two gases, ammonia and hydrogen chloride, which is denser? Name the method of collection of this gas.
- Of the two gases, ammonia and hydrogen chloride, which is more dense? Name the method of collection of this gas.
- Give one example of a reaction between the above two gases which produce a solid compound.
- Write the balanced equation for a reaction in which ammonia is oxidized by:
- A metal oxide
- a gas which is not oxygen
