Advertisements
Advertisements
Question
Estimate the average thermal energy of a helium atom at the temperature on the surface of the Sun (6000 K).
Solution
On the surface of the sun, T = 6000 K
Average thermal energy = `3/2` kT
`= 3/2 xx 1.38 xx 10^(-38) xx 6000`
`= 1.241 xx 10^(-19)` J
Hence, the average thermal energy of a helium atom on the surface of the sun is 1.241 × 10–19 J.
APPEARS IN
RELATED QUESTIONS
The figure shows the plot of PV/T versus Pfor 1.00×10–3 kg of oxygen gas at two different temperatures.
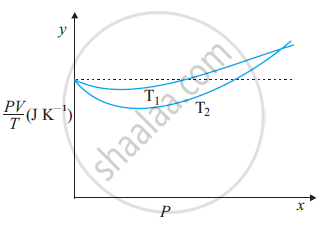
(a) What does the dotted plot signify?
(b) Which is true: T1 > T2 or T1 < T2?
(c) What is the value of PV/T where the curves meet on the y-axis?
(d) If we obtained similar plots for 1.00 ×10–3 kg of hydrogen, would we get the same value of PV/T at the point where the curves meet on the y-axis? If not, what mass of hydrogen yields the same value of PV/T (for low pressure high temperature region of the plot)? (Molecular mass of H2 = 2.02 u, of O2 = 32.0 u, R = 8.31 J mo1–1 K–1.)
An oxygen cylinder of volume 30 litres has an initial gauge pressure of 15 atm and a temperature of 27 °C. After some oxygen is withdrawn from the cylinder, the gauge pressure drops to 11 atm and its temperature drops to 17 °C. Estimate the mass of oxygen taken out of the cylinder (R = 8.31 J mol–1 K–1, molecular mass of O2 = 32 u)
Give reasons for the following:
Gas fills the vessel completely in which it is kept.
Fill in the blanks:
If the temperature is reduced to half, ………….. would also reduce to half.
The average energy per molecule is proportional to ______
If the absolute temperature of a body is doubled, the power radiated will increase by a factor of ______
Show that for diatomic gas the ratio of the two specific heats is 7:5.
Estimate the average thermal energy of a helium atom at room temperature (27 °C).
A box of 1.00 m3 is filled with nitrogen at 1.50 atm at 300K. The box has a hole of an area 0.010 mm2. How much time is required for the pressure to reduce by 0.10 atm, if the pressure outside is 1 atm.
P ∝ T at constant volume is the statement of ______.
