Advertisements
Advertisements
Question
Estimate the average thermal energy of a helium atom at room temperature (27 °C).
Solution
At room temperature, T = 27°C = 300 K
Average thermal energy = `3/2` kT
Where k is Boltzmann constant = 1.38 × 10–23 m2 kg s–2 K–1
`therefore 3/2kT = 3/2 xx 1.38 xx 10^(-38) xx 300`
= 6.21 × 10–21J
Hence, the average thermal energy of a helium atom at room temperature (27°C) is 6.21 × 10–21 J.
APPEARS IN
RELATED QUESTIONS
Estimate the total number of air molecules (inclusive of oxygen, nitrogen, water vapour and other constituents) in a room of capacity 25.0 m3 at a temperature of 27 °C and 1 atm pressure
50 m3 of saturated vapour is cooled down from 30°C to 20°C. Find the mass of the water condensed. The absolute humidity of saturated water vapour is 30 g m−3 at 30°C and 16 g m−3 at 20°C.
What do you understand by gas?
Give reasons for the following:
Gas fills the vessel completely in which it is kept.
Match the following:
|
|
Column A |
Column B |
|
(a) |
cm3 |
(i) Pressure |
|
(b) |
Kelvin |
(ii) Temperature |
|
(c) |
Torr |
(iii) Volume |
|
(d) |
Boyle's law |
(iv) `"V"/"T" = ("V"_1)/("T"_1)` |
|
(a) |
Charles's law |
(v) `"PV"/"T" = ("P"_1 "V"_1)/"T"_1` |
|
|
|
(vi) PV = P1V1 |
Name or state the following:
An equation used in chemical calculations which gives a simultaneous effect of changes of temperature and pressure on the volume of a given mass of dry gas
Give reason for the following:
Volumes of gases are converted into s.t.p. conditions and then compared.
Show that for monoatomic gas the ratio of the two specific heats is 5:3.
Which of the following diagrams (Figure) depicts ideal gas behaviour?
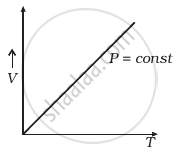 (a) |
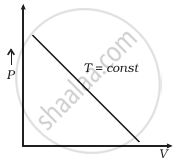 (b) |
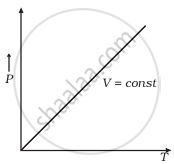 (c) |
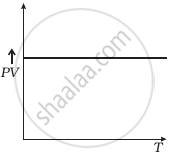 (d) |
P ∝ T at constant volume is the statement of ______.
