Advertisements
Advertisements
Question
Estimate the total number of air molecules (inclusive of oxygen, nitrogen, water vapour and other constituents) in a room of capacity 25.0 m3 at a temperature of 27 °C and 1 atm pressure
Solution 1
Volume of the room, V = 25.0 m3
Temperature of the room, T = 27°C = 300 K
Pressure in the room, P = 1 atm = 1 × 1.013 × 105 Pa
The ideal gas equation relating pressure (P), Volume (V), and absolute temperature (T) can be written as:
PV = kBNT
Where,
KB is Boltzmann constant = 1.38 × 10–23 m2 kg s–2 K–1
N is the number of air molecules in the room
`:. N = (PV)/(k_BT)`
`= (1.013xx10^5xx25)/(1.38xx10^(-23)xx300) = 6.11 xx 10^(26)` molecules
Therefore, the total number of air molecules in the given room is 6.11 × 1026.
Solution 2
Here, Volume of room, `V= 25.0 m^3`, temperature, `T = 27 ^@C = 300 K` and
Pressure, `P = 1 "atm" = 1.01 xx 10^5 Pa`
According to gas equation, `PV = muRT = muN_A.k_BT`
Hence, total number of air molecules in the volume of given gas
`N = mu.N_A = PV/K_BT`
`:. N = (1.01 xx 10^5 xx25.0)/((1.38 xx 10^(-23))xx300) = 6.1 xx 10^(26)`
APPEARS IN
RELATED QUESTIONS
Molar volume is the volume occupied by 1 mol of any (ideal) gas at standard temperature and pressure (STP: 1 atmospheric pressure, 0 °C). Show that it is 22.4 litres
Three vessels of equal capacity have gases at the same temperature and pressure. The first vessel contains neon (monatomic), the second contains chlorine (diatomic), and the third contains uranium hexafluoride (polyatomic).
Is the root mean square speed of molecules the same in the three cases? If not, in which case is vrms the largest?
At what temperature is the root mean square speed of an atom in an argon gas cylinder equal to the rms speed of a helium gas atom at – 20 °C? (atomic mass of Ar = 39.9 u, of He = 4.0 u).
50 m3 of saturated vapour is cooled down from 30°C to 20°C. Find the mass of the water condensed. The absolute humidity of saturated water vapour is 30 g m−3 at 30°C and 16 g m−3 at 20°C.
Give reasons for the following:
Gas fills the vessel completely in which it is kept.
Name or state the following:
The absolute temperature value corresponding to 35°C.
If the absolute temperature of a body is doubled, the power radiated will increase by a factor of ______
Which of the following diagrams (Figure) depicts ideal gas behaviour?
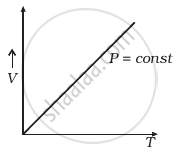 (a) |
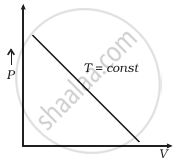 (b) |
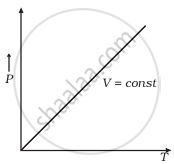 (c) |
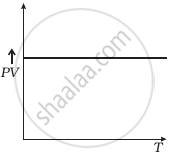 (d) |
P ∝ T at constant volume is the statement of ______.
Two tanks of equal volume contain equal mass of oxygen and nitrogen at 127°C. Find the ratio of pressure in two tanks.
