Advertisements
Advertisements
Question
Explain [Fe(CN)6]3− is an inner orbital complex, whereas [FeF6]3− is an outer orbital complex.
[Atomic number: Fe = 26]
Solution
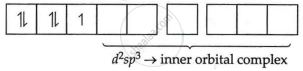
[Fe(CN)6]3−
Fe = 3 oxidation state
i.e., electronic configuration = 3d5
CNΘ = strong field ligand
i.e., Δ0 > P pairing will take place
Electronic configuration of Fe3+ = 3d5
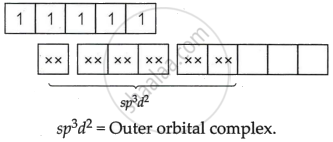
[FeF6]3−
Fe = +3 oxidation state
F− = Weak field ligand Δ0 < P `->` Pairing will not take place.
APPEARS IN
RELATED QUESTIONS
For the complex [Fe(H2O)6]+3, write the hybridisation, magnetic character and spin of the complex. (At, number : Fe = 26)
Write the hybridization and magnetic behaviour of the complex [Ni(CO)4].
(At.no. of Ni = 28)
Write the hybridization and shape of the following complexe : [CoF6]3–
(Atomic number : Co = 27, Ni = 28)
Explain [Co(NH3)6]3+ is an inner orbital complex whereas [Ni(NH3)6]2+ is an outer orbital complex.
Write down the IUPAC name of the following complex and indicate the oxidation state, electronic configuration and coordination number. Also, give the stereochemistry and magnetic moment of the complex:
K[Cr(H2O)2(C2O4)2].3H2O
Write the hybridization and magnetic character of the following complexes:
[Fe(H2O)6]2+
(Atomic no. of Fe = 26)
Write the hybridization and magnetic character of the following complexes:
[Fe(CO)5]
(Atomic no. of Fe = 26)
Magnetic moment of \[\ce{[MnCl4]^{2-}}\] is 5.92 BM. Explain giving reason.
Why do compounds having similar geometry have different magnetic moment?
Assertion: \[\ce{[Fe(CN)6]^{3-}}\] ion shows magnetic moment corresponding to two unpaired electrons.
Reason: Because it has d2sp3 type hybridisation.
