Advertisements
Advertisements
Question
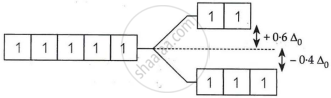
On the basis of crystal field theory, write the electronic configuration for the d5 ion with a weak ligand for which Δ0 < P.
Solution
For weak field ligand Δ0 < P i.e., pairing will not occur for d5 ion.
RELATED QUESTIONS
On the basis of crystal field theory, write the electronic configuration for d4 ion if Δ0 > P.
The hexaquo manganese (II) ion contains five unpaired electrons, while the hexacyanoion contains only one unpaired electron. Explain using Crystal Field Theory.
Why are low spin tetrahedral complexes rarely observed?
Complete and balance the following reactions:
(1) P4 + H2SO4 → ____ + _____ + _____ + _____
(2) Ag + HNO3(dilute) → _____ + ______ + _____ + _____
Using crystal field theory, draw energy level diagram, write electronic configuration of the central metal atom/ion and determine the magnetic moment value in the following:
\[\ce{[CoF6]^{3-}, [Co(H2O)6]^{2+}, [Co(Cn)6]^{3-}}\]
Using crystal field theory, draw energy level diagram, write electronic configuration of the central metal atom/ion and determine the magnetic moment value in the following:
\[\ce{[FeF6]^{3-}, [Fe(H2O)6]^{2+}, [Fe(CN)6]^{4-}}\]
Consider that d6 metal ion (M2+) forms a complex with aqua ligands and the spin only magnetic moment of the complex is 4.90 BM. The geometry and the crystal field stabilization energy of the complex is:
On the basis of Crystal Field theory, write the electronic configuration for the d5 ion with a strong field ligand for which Δ0 > P.
On the basis of crystal field theory, write the electronic configuration for d4 with a strong field ligand for which Δ0 > P.
Explain the difference between a weak field ligand and a strong field ligand.
