Advertisements
Advertisements
Question
For the following reactions
- \[\ce{CH3CH2CH2Br + KOH → CH3 – CH = CH2 + KBr + H2O}\]
- \[\ce{(CH3)3CBr + KOH → (CH3)3COH + KBr}\]
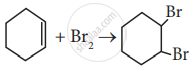
Which of the following statement is correct?
Options
(A) is elimination, (B) and (C) are substitution
(A) is substitution, (B) and (C) are elimination
(A) and (B) are elimination and (C) is addition reaction
(A) is elimination, B is substitution and (C) is addition reaction.
Solution
(A) is elimination, B is substitution and (C) is addition reaction.
APPEARS IN
RELATED QUESTIONS
What is the hybridisation state of benzyl carbonium ion?
Which of the following species is not electrophilic in nature?
Homolytic fission of covalent bond leads to the formation of ______.
Hyper Conjugation is also known as
Write a short note on hyperconjucation.
What is electrophiles? Give a suitable example.
What is nucleophiles? Give a suitable example.
Show the heterolysis of a covalent bond by using curved arrow notation and complete the following equation. Identify the nucelophile.
CH3 – Br + KOH →
Show the heterolysis of a covalent bond by using curved arrow notation and complete the following equation. Identify the nucelophile.
\[\ce{CH3 – O – CH3 + HI →}\]
Give example for the following type of organic reaction.
β - elimination
