Advertisements
Advertisements
Question
Give appropriate scientific reasons for Zinc oxide can be reduced to zinc metal by using
carbon, but aluminium oxide cannot be reduced by a reducing agent
Solution
The metals in the middle of the activity series like zinc are moderately reactive, and carbon is a good reducing agent because of which zinc oxide gets easily reduced by carbon. Oxides of highly active metals like aluminium have the great affinity towards oxygen and so cannot be reduced by carbon
APPEARS IN
RELATED QUESTIONS
An element L consists of molecules.
When L is heated with iron metal, it forms a compound FeL. What chemical term would you use to describe the change undergone by L?
Give reason for the following:
Carbon anodes are used in the electrolytic extraction of aluminium.
Name oxide of one metal which is reduced by (give equation): Reduction with heat alone
For the reaction of a metal oxide. Suggest a reducing agent other than carbon
4 tones of bauxite, 150 Kg of sodium hydroxide and 600 Kg of graphite.
The aluminium compound in bauxite is aluminium oxide and the main impurity is iron (II) oxide. Aluminium is obtained by the electrolysis of aluminium oxide dissolved in cryolite.
(a) When bauxite is treated with sodium hydroxide solution, what happens to:
(i) the aluminium oxide?
(ii) The iron (III) oxide?
In order to obtain one tone of aluminium, the following inputs are required:
4 tones of bauxite, 150 Kg of sodium hydroxide and 600 Kg of graphite.
The aluminium compound in bauxite is aluminium oxide and the main impurity is iron (II) oxide. Aluminium is obtained by the electrolysis of aluminium oxide dissolved in cryolite.
(i) Write the formula of cryolite.
(ii) Write down the word which correctly completes the following sentence.
"By dissolving aluminium oxide in cryolite, a ______ (conducting / non- conducting) solution is produced.
(iii) Why so much graphite required for this electrolytic process.
(iv) Write the equation for the reaction which takes place at cathode.
Name the following:
The compound formed by the reaction between calcium oxide and silica.
Complete the following by selecting the correct option from the choices given :
The diavalent metal whose oxide is reduced to metal by electrolysis of its fused salt is ________
Write the observation for the following:
A paper dipped in potassium permanganate solution is put on the mouth of a test tube containing sulphur dioxide gas.
The following sketch represents the electroplating of an Iron cup with Nickel metal.
Study the diagram and answer the following questions:
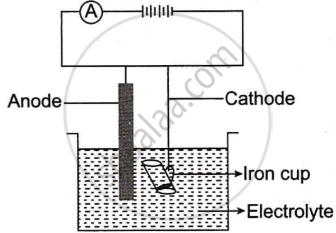
- During electroplating, the iron cup is placed at the cathode. Why?
- Name the ion that must be present in the electrolyte.
- State one condition that is necessary to ensure that the deposit is smooth, firm and even.
- Write the reaction taking place at the cathode.
- What change would you observe at the anode?
