Advertisements
Advertisements
Question
Name oxide of one metal which is reduced by (give equation): Reduction with heat alone
Solution
Mercury
\[\ce{ 2HgO ->[Heat] 2Hg + O2}\]
APPEARS IN
RELATED QUESTIONS
Name the compound added to pure alumina to lower the fusion temperature during the electrolytic reduction of alumina.
How is the following metallic oxide reduced? Write equations:
Iron (ll) oxide
Give reason for the following:
Carbon anodes are used in the electrolytic extraction of aluminium.
Give equation for the following conversion: Ferric oxide to iron.
Give equation for the following conversion: Aluminium hydroxide to aluminium oxide.
For the reaction of a metal oxide. Suggest a reducing agent other than carbon
what is the function of cryolite in the extraction of aluminium, other than acting as a solvent for bauxite?
Name a non-metallic element which is a conductor of electricity.
Answer the following question :
Name the process used for the enrichment of sulphide ore.
The following sketch represents the electroplating of an Iron cup with Nickel metal.
Study the diagram and answer the following questions:
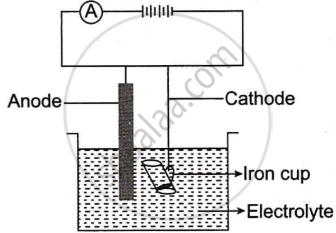
- During electroplating, the iron cup is placed at the cathode. Why?
- Name the ion that must be present in the electrolyte.
- State one condition that is necessary to ensure that the deposit is smooth, firm and even.
- Write the reaction taking place at the cathode.
- What change would you observe at the anode?
