Advertisements
Advertisements
Question
Give reason Aromatic diazonium salts are more stable than aliphatic diazonium salts.
Solution 1
Due to resonance, there is a dispersal of positive charge on the benzene ring. This resonance accounts for the stability of the diazonium ion. Hence, diazonium salts of aromatic amines are more stable than those of aliphatic amines.
shaalaa.com
Solution 2
Aromatic diazonium salts are stable due to the dispersed of positive charge over the benzene ring
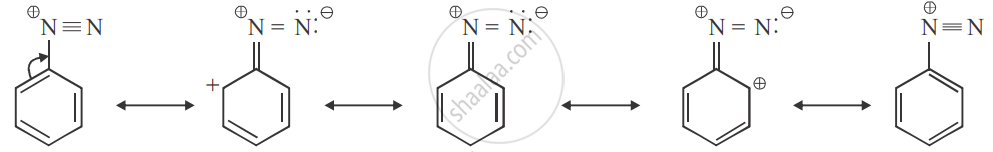
Dispersal of charge is not possible in aliphatic diazonium salts.
shaalaa.com
Is there an error in this question or solution?
