Advertisements
Advertisements
Question
Give scientific reasons for the following:
Bottled drinks are cooled more effectively when surrounded by lumps of ice than by cold water at 0°.
Solution 1
Lumps of ice cool better than cold water because each gram of ice requires additional 80 calories of heat to get converted into water. Hence, cooling capacity of lumps of ice is more than cold water.
Solution 2
Every 1 kg of ice at 0°C absorbs 336,000 J of heat energy to form water at 0°C. As ice can extract 336,000 J of heat energy, more than water at 0°C, therefore it cools the bottled drinks more effectively.
RELATED QUESTIONS
How can a temperature in degree Celsius be converted into S.I. unit of temperature?
Name the law on which the principle of mixture is based.
How will you determine the specific heat capacity of a liquid like olive oil by the method of mixtures?
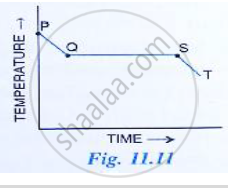
Fig 11. 11 shows the variation in temperature with time when some wax cools from the liquid phase to the solid phase.
(i) In which part of the curve, the wax is in liquid phase?
(ii) What does the part QS of the curve represent?
(iii) In which part of the curve, the wax will be the in the liquid as well as solid phase?
(iv) In which part of the curve, the wax is in solid phase?
1 kg of ice at 0°C is heated at a constant rate and its temperature is recorded after every 30 s till steam is formed at 100℃. Draw a temperature time graph to represent the change of phase.
The amount of heat energy required to convert 1 kg of ice at – 10℃ to water at 100℃ is 7,77,000 J. Calculate the specific latent heat of ice. Specific heat capacity of ice = 2100 J kg-1K-1, Specific heat capacity of water = 4200 J kg-1 K-1.
Which requires more heat: 1 g ice at 0°C or 1 g water at 0°C to raise its temperature to 10°C? Explain your answer.
What energy change would you Expect to take place in the molecules of a substance when it undergoes:
(i) a change in its temperature?
(ii) a change in itsstate without any change in its temperature?
