Advertisements
Advertisements
Question
Give a simple chemical test to distinguish between Aniline and N, N-dimethylaniline
Solution 1
Aniline is a primary amine while N, N-dimethylaniline is a tertiary amine. They both can be distinguished by Carbylamine test. Primary amines on heating with chloroform and ethanolic potassium hydroxide, form foul-smelling isocyanides or carbylamines. Aniline, being an aromatic primary amine gives positive carbylamine test. However, N, N-dimethylaniline being a tertiary amine does not.
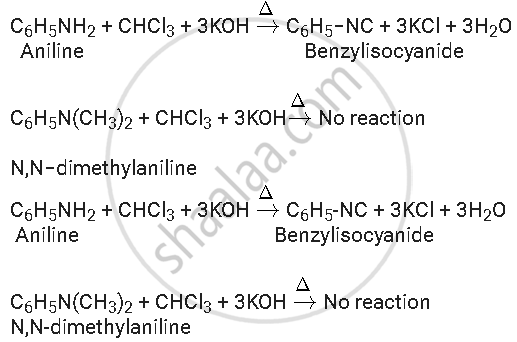
Solution 2
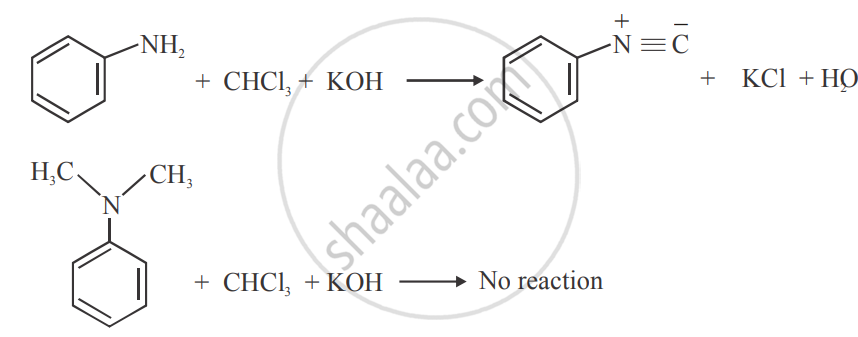
Carbylamine test can be used to distinguish.
APPEARS IN
RELATED QUESTIONS
Which of the following amines yields foul smelling product with holoform and alcoholic KOH?
(a) Ethyl amine
(b) Diethyl amine
(c) Triethyi amine
(d) Ethyl methyl amine
Write the chemical equations involved in the following reactions:
Carbylamine reaction
Write the chemical equations involved when aniline is treated with the following reagents:
CHCI3 + KOH
Write equations of the following reactions:
Carbyl amine reaction
Which one of the following shows the carbylamine reaction in the presence of heat?
A primary amine, \[\ce{RNH2}\] can be reacted with \[\ce{CH3-X}\] to get secondary amine, \[\ce{R-NHCH3}\] but the only disadvantage is that 3° amine and quaternary ammonium salts are also obtained as side products. Can you suggest a method where \[\ce{RNH2}\] forms only 2° amine?
Assertion: Acetanilide is less basic than aniline.
Reason: Acetylation of aniline results in decrease of electron density on nitrogen.
What happens when reactions:
Aniline reacts with chloroform in the presence of alcoholic potassium hydroxide
Aniline and diphenylamine may be distinguished by ______.
The reaction of a primary amine with chloroform and ethanolic KOH is called ______.
