Advertisements
Advertisements
Question
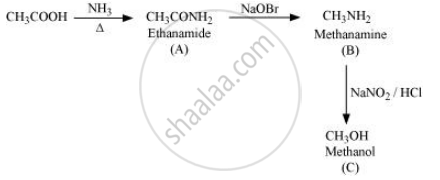
Give the structures of A, B and C in the following reaction:
Solution
APPEARS IN
RELATED QUESTIONS
Identify the compounds 'A' and 'B' in the following equation:

An aromatic compound 'A' of molecular formula C7H7ON undergoes a series of reactions as shown below. Write the structures of A, B, C, D and E in the following reactions :

Why cannot aromatic primary amines be prepared by Gabriel phthalimide synthesis?
Explain the mechanism of action of hydroiodic acid on 3-methylbutan-2-ol.
Write the order of reactivity of alkyl halides with ammonia.
\[\ce{CH3-CN ->[Na/C2H5OH]}\]
The product formed is ____________.
Which of the following amines forms a clear solution when treated with benzene sulphonyl chloride and excess of potassium hydroxide?
Identify the product obtained when benzamide is treated with bromine and aqueous sodium hydroxide?
Which of the following reagents is used in Mendius reduction reaction of alkyl cyanide?
Identify product B in the following reaction.
\[\ce{Aniline ->[NaNO2][HCl] A ->[KI] B}\]
Which of the following reagents would not be a good choice for reducing an aryl nitro compound to an amine?
The source of nitrogen in Gabriel synthesis of amines is ______.
Hoffmann Bromamide Degradation reaction is shown by ______.
Write following conversions:
acetanilide `->` p-nitroaniline
Assertion: Hoffmann’s bromamide reaction is given by primary amines.
Reason: Primary amines are more basic than secondary amines.
Describe Gabriel's phthalimide synthesis. (Give reaction)
Identify A and B in the following reaction.
\[\ce{C6H5CH2Br ->[Alco.][KCN] A ->[Na/Ethanol][reduction] B}\]
Write short notes on the following:
Ammonolysis
