Advertisements
Advertisements
Question
How are proteins classified on the basis of molecular shapes?
Solution
On the basis of molecular shape, proteins are classified into two types—fibrous proteins and globular proteins.
- Fibrous proteins:- They are insoluble in water, long, thread-like and tend to lie side by side to form fibres. The polypeptide chains are held together by hydrogen bonds. Examples: Collagen in tendons; keratin in hair, skin, nails, horn and feathers; myosin in muscle; fibroin in silk
- Globular proteins:- They are soluble in water and aqueous solutions of bases, acids and salts. They are folded to form a spherical shape, have intramolecular hydrogen bonding and have weak intermolecular forces as compared to fibrous proteins.
APPEARS IN
RELATED QUESTIONS
How is tripeptide formed?
Define the following as related to proteins
Peptide linkage
Differentiate between the following :
Fibrous proteins and Globular proteins
Write one difference between α-helix and β-pleated structures of proteins.
Differentiate between the following :
Peptide linkage and Glycosidic linkage
Which of the following biomolecules is insoluble in water?
The protein responsible for blood clotting is ____________.
The helical structure of protein is stabilized by:
The correct statement for protein haemoglobin.
Which of the following statement is correct:
Proteins are found to have two different types of secondary structures viz. α-helix and β-pleated sheet structure. α-helix structure of protein is stabilised by:
Dinucleotide is obtained by joining two nucleotides together by phosphodiester linkage. Between which carbon atoms of pentose sugars of nucleotides are these linkages present?
Optical rotations of some compounds along with their structures are given below which of them have D configuration.
| (I) | 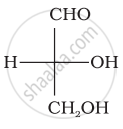 |
| (II) | 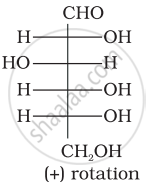 |
| (III) | 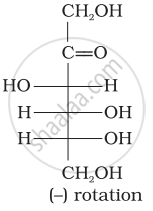 |
Proteins can be classified into two types on the basis of their molecular shape i.e., fibrous proteins and globular proteins. Examples of globular proteins are:
(i) Insulin
(ii) Keratin
(iii) Albumin
(iv) Myosin
In fibrous proteins, polypeptide chains are held together by:
(i) van der Waals forces
(ii) disulphide linkage
(iii) electrostatic forces of attraction
(iv) hydrogen bonds
Structures of glycine and alanine are given below. Show the peptide linkage in glycylalanine.
\[\ce{\underset{(Glycine)}{H2N - CH2 - COOH}}\];
\[\begin{array}{cc}
\ce{H2N - CH2 - COOH}\\
|\phantom{......}\\
\ce{\underset{(Alanine)}{CH3}\phantom{...}}
\end{array}\]
Which moieties of nucleosides are involved in the formation of phosphodiester linkages present in dinucleotides? What does the word diester in the name of linkage indicate? Which acid is involved in the formation of this linkage?
Assertion: β-glycosidic linkage is present in maltose,

Reason: Maltose is composed of two glucose units in which C–1 of one glucose unit is linked to C–4 of another glucose unit.
Each polypeptide in a protein has amino acids linked with each other in a specific sequence. This sequence of amino acids is said to be ______.
Peptide linkage is:
Explain formation of peptide linkage in protein with an example.
Proteins are polymers of ______.
Assertion (A): Proteins are polymers of α-amino acids connected by a peptide bond.
Reason (R): A tetrapeptide contains 4 amino acids linked by 4 peptide bonds.
β-pleated sheet structure in proteins refers to ______.
Write a classification of proteins with an example.
