Advertisements
Advertisements
Question
How can the following conversions be brought about:
(i) Acetic acid to methyl cyanide.
(ii) Acetaldehyde to formaldehyde.
(iii) Nitrobenzene to 2, 4, 6 tribromoaniline.
Solution
(i)
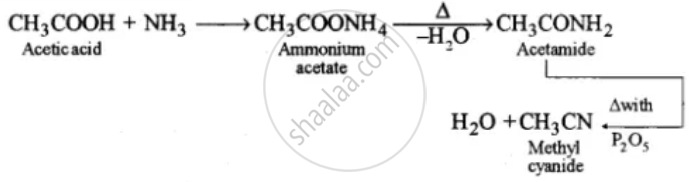
(ii)
\[\ce{\underset{\text{Acetaldehyde}}{CH3CHO} ->[(O)]\underset{\text{Acetic acid}}{CH3COOH}->[+NH3][\Delta(-H2O)] +\underset{\text{Acetanide}}{CH3CONH2}->[Br2/KOH]\underset{\text{Formaldehyde}}{HCHO}<-[(O)]\underset{\text{Methyl alcohol}}{CH3OH}<-[NaNO2 + HCl]\underset{\text{Methylamine}}{CH3NH2}}\]
(iii)

APPEARS IN
RELATED QUESTIONS
How can the following conversions be brought about:
Chlorobenzene to phenol
A solution of sodium chloride has no effect on litmus, but a solution of zinc chloride turns blue litmus red.
Identify the reagents A, B, C, D, E and F required for the following conversion:
\[\ce{C6H5NO2->[A]C6H5NH2->[B]C6H5N2+Cl- ->[C]C6H5Cl->[D]C6H5OH->[H2SO4]E + F}\]
Complete the following reaction:
\[\begin{array}{cc}
\ce{CH3}\phantom{..............}\\
|\phantom{..............}\phantom{...}\\
\ce{CH3-CH-COOH->[(i) Br2/Red P4][(ii) H2O]}\\
\phantom{.......}\
\end{array}\]
The acid which doesn't contain COOH group is
Formic acid when heat with H2SO4 gives
The oxidation of toluene with hot KMnO4 gives ______.
Why formic acid is stronger than acetic acid?
Carboxylic acid present in curd is ______.
