Advertisements
Advertisements
Question
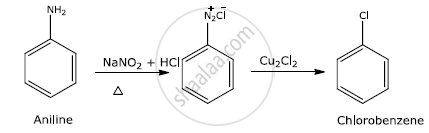
How is chlorobenzene prepared from aniline?
Solution
Aniline reacts with nitrous acid to give benzene diazonium chloride which on treatment with cuprous chloride gives chlorobenzene
APPEARS IN
RELATED QUESTIONS
What is the action of acetic anhydride on ethylamine?
What is the action of the following reagents on aniline?
Bromine water
Give reasons for the following:
Aniline does not undergo Friedel- Crafts reaction.
Write the structures of main products when aniline reacts with the following reagents :
Br2 water
Write the structures of main products when aniline reacts with the following reagents :
(CH3CO)2O/pyridine
Write the chemical equations involved when aniline is treated with the following reagents:
Br2 water
Account for the following:
Although the amino group is o, p-directing in aromatic electrophilic substitution reactions, aniline on nitration gives a substantial amount of m-nitroaniline.
Write short notes on Acetylation
What is the action of acetic anhydride on diethylamine?
What is the action of the following reagents on aniline?
Acetic anhydride
Illustrate the following reactions giving suitable example in each case
Acetylation of amines
How will you convert the following?
Aniline into N−phenylethanamide
In the nitration of benzene using a mixture of conc. \[\ce{H2SO4}\] and conc. \[\ce{HNO3}\], the species which initiates the reaction is ______.
Assertion: N, N-Diethylbenzene sulphonamide is insoluble in alkali.
Reason: Sulphonyl group attached to nitrogen atom is strong electron-withdrawing group.
When bromination of aniline is carried out by protecting – NH2. The major product is
Give reasons for the following observation:
Aniline is acetylated before nitration reaction.
Give reasons for the following observation:
Aniline does not react with methyl chloride in the presence of anhydrous AlCl3 catalyst.
In the following reaction, the reason for why meta-nitro product also formed is:

How can the activating effect of the −NH2 group in aniline be controlled?
