Advertisements
Advertisements
Question
How is sodium metal extracted? Explain with the help of equation of the reaction involved.
Solution
Sodium is extracted by electrolytic reduction of molten sodium chloride. On passing electricity through molten sodium chloride, decomposition occurs and sodium metal and chlorine gas are formed.
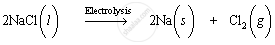
Molten sodium chloride contains sodium and chloride ions. The reactions that occur during electrolysis are:
1. The cathode produces electrons to reduce sodium ions to sodium atoms (or sodium metal) by acting as reducing agent. Sodium ions are cations; therefore, they are attracted to the negatively-charged cathode and get deposited there.
Cathode: 
Chloride ions are anions; therefore they are attracted to the positively-charged anode. These chloride ions are oxidised to chlorine gas. Chlorine gas is produced at the anode.
Anode: 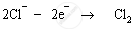
APPEARS IN
RELATED QUESTIONS
What chemical process is used for obtaining a metal from its oxide?
A zinc ore gave CO2 on treatment with a dilute acid. Identify the ore and write its chemical formula.
Name one ore of copper (other than cuprite). Which compound of copper is present in this ore? Also, write its chemical formula.
Which metal is extracted from calamine ore?
Which metal can be extracted from the following ore?
Haematite
Name the following:
Two metals which are found in the free or native state.
Name the following:
A compound which is added to lower the fusion temperature of the electrolytic bath in the extraction of aluminium.
Name the methods by which concentrated ore is converted to metallic oxide.
The process in which a carbonate ore is heated strongly in the absence of air to convert it into metal oxide is called ____________.
The given reaction shows one of the processes to extract the metals like Iron and Manganese.
\[\ce{MnO2(s) + Al(s) -> Mn(l) + Al2O3(s) + Heat}\]
Give the reason why the above reaction is known as a thermite reaction.
