Advertisements
Advertisements
Question
\[\ce{H2O2}\] is a better oxidising agent than water. Explain.
Solution
\[\ce{H2O2}\] is a better oxidizing agent than water. \[\ce{H2O2}\] acts as an oxidizing agent in acid as well as in alkaline medium.
\[\ce{H2O2 + 2H^{+} + 2e^{-} ->[Acid] 2H2O; E^Θ = 1.77 V}\]
\[\ce{H2O2 + OH^{-} + 2e^{-} ->[Alkaline] 3OH-; E^Θ = 0.88 V}\]
Oxidation state of oxygen changes from – 1 to – 2. Oxidising nature of \[\ce{H2O2}\] can be interpreted on account of possession of labile oxygen.
\[\ce{H2O2 -> H2O + O}\]
When water acts as an oxidizing agent, it is reduced to \[\ce{H2}\]. Water reacts with number of active metals whose electrode potential is less than – 0.83 V.
\[\ce{\underset{Reduction}{2Na} + \underset{Oxidant}{2H2O} -> 2NaOH + H2}\]
APPEARS IN
RELATED QUESTIONS
Why is it said that-
Water is a universal solvent.
Explain the picture in your own words.
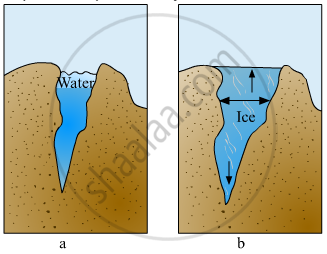
Explain the various properties of water.
Why is the density of seawater more than that of rain water?
Which of the following equations depict the oxidising nature of \[\ce{H2O2}\]?
Give reasons: Ice floats on water.
Why does water show high boiling point as compared to hydrogen sulphide? Give reasons for your answer.
Freezing of water will cause an ______ is the volume.
What is the taste of distilled water & boiled water?
What are the physical properties of pure water?
