Advertisements
Advertisements
Question
If NaCl is doped with 10−3 mol % of SrCl2, what is the concentration of cation vacancies?
Solution
It is given that NaCl is doped with 10−3 mol% of SrCl2.
This means that 100 mol of NaCl is doped with 10−3 mol of SrCl2.
Therefore, 1 mol of NaCl is doped with `10^(-3)/100` mol of SrCl2
= 10−5 mol of SrCl2
Cation vacancies produced by one Sr2+ ion = 1
Concentration of the cation vacancies Produced by 10-5 mol of Sr2+ ions = `10^(-5)xx6.022xx10^(23)`
=6.022 x 1018 mol-1
Hence, the concentration of cation vacancies created by SrCl2 is 6.022 × 108 per mol of NaCl.
APPEARS IN
RELATED QUESTIONS
Explain the following terms with suitable examples: F-centres
Yellow colour of NaCl crystals in sodium vapour is due to ____________.
The following diagram shows:
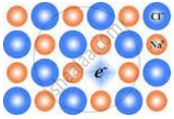
Which of the following will have metal deficiency defect?
Which of the following is a non-stoichiometric defect?
Pink colour of LiCl crystals is due to ______.
Why does table salt, NaCl, some times appear yellow in colour?
Why does white ZnO (s) becomes yellow upon heating?
Match the types of defect given in Column I with the statement given in Column II.
| Column I | Column II |
| (i) Impurity defect | (a) NaCl with anionic sites called F-centres |
| (ii) Metal excess defect | (b) FeO with Fe3+ |
| (iii) Metal deficiency defect | (c) NaCl with Sr2+ and some cationic sites vacant |
A sample of ferrous oxide has actual formula Fe0.93O1.00. In this sample what fraction of metal ions are Fe2+ ions? What type of nonstoichiometric defect is present in this sample?
The appearance of colour in solid alkali metal halides is generally due to
On doping Ge metal with a little of In (indium), we get:
The non-stoichiometric compound Fe0 940 is formed when x% of Fe2+ ions are replaced by as many `2/3` Fe3+ ions. The value of x is
Non-stoichiometric defects refer to ______
