Advertisements
Advertisements
Question
If water contains dissolved calcium hydrogen carbonate, out of soaps and synthetic detergents which one will you use for cleaning clothes?
Solution 1
Synthetic detergents are preferred for cleaning clothes. When soaps are dissolved in water containing calcium ions, these ions form insoluble salts that are of no further use.
However, when synthetic detergents are dissolved in water containing calcium ions, these ions form soluble salts that act as cleansing agents.
Solution 2
Calcium hydrogencarbonate makes water hard. Therefore, soap cannot be used because it gets precipitated in hard water. On the other hand, a synthetic detergent does not precipitate in hard water because its calcium salt is also soluble in water. Therefore, synthetic detergents can be used for cleaning clothes in hard water.
APPEARS IN
RELATED QUESTIONS
How are synthetic detergents better than soap?
What are biodegradable and non-biodegradable detergents? Give one example of each.
Label the hydrophilic and hydrophobic parts in the following compounds.
`CH_3(CH_2)_16COO(CH_2CH_2O)_nCH_2CH_2OH`
Define disinfectants with a Suitable example?
Which of the following is an example of liquid dishwashing detergent?
Polyethyleneglycols are used in the preparation of which type of detergents?
Explain why some times foaming is seen in river water near the place where sewage water is poured after treatment?
Hair shampoos belong to which class of synthetic detergent?
Draw the diagram showing micelle formation by the following detergent.
\[\ce{CH3(CH2)10CH2OS\overset{-}{O}3\overset{+}{N}a}\]
How does the branching of hydrocarbon chain of synthetic detergents affect their biodegradability?
Match structures given in Column I with the type of detergents given in Column II.
| Column I | Column II |
| (i) \[\ce{CH3(CH2)16COO(CH2CH2O) nCH2CH2OH}\] | (a) Cationic detergent |
| (ii) \[\ce{C17H35COO- Na+}\] | (b) Anionic detergent |
| (iii) \[\ce{CH3-(CH2)10CH2SO3- Na+}\] | (c) Nonionic detergent |
|
(iv) |
(d) Soap |
Synthetic detergents have advantage over usual soaps as far as cleansing power is concerned. But use of synthetic detergents over a long time creates environmental pollution. How can the pollution caused by synthetic detergents be minimised? Classify the detergents according to their chemical nature.
Define the following:
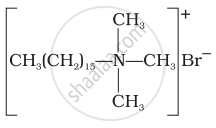
Cationic detergents
Explain the following term with suitable examples.
cationic detergents
Explain the following term with a suitable example.
cationic detergents
Explain the Following Term with Suitable Examples.
Cationic Detergents