Advertisements
Advertisements
Question
Match structures given in Column I with the type of detergents given in Column II.
| Column I | Column II |
| (i) \[\ce{CH3(CH2)16COO(CH2CH2O) nCH2CH2OH}\] | (a) Cationic detergent |
| (ii) \[\ce{C17H35COO- Na+}\] | (b) Anionic detergent |
| (iii) \[\ce{CH3-(CH2)10CH2SO3- Na+}\] | (c) Nonionic detergent |
|
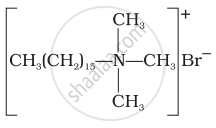
(iv) |
(d) Soap |
Solution
| Column I | Column II |
| (i) \[\ce{CH3(CH2)16COO(CH2CH2O) nCH2CH2OH}\] | (c) Nonionic detergent |
| (ii) \[\ce{C17H35COO- Na+}\] | (d) Soap |
| (iii) \[\ce{CH3-(CH2)10CH2SO3- Na+}\] | (b) Anionic detergent |
|
(iv) |
(a) Cationic detergent |
APPEARS IN
RELATED QUESTIONS
Explain the following terms with suitable examples - Anionic detergents
Explain the following terms with suitable examples - Non-ionic detergents
If water contains dissolved calcium hydrogen carbonate, out of soaps and synthetic detergents which one will you use for cleaning clothes?
Label the hydrophilic and hydrophobic parts in the following compounds.
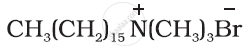
Which of the following statements are correct?
(i) Cationic detergents have germicidal properties.
(ii) Bacteria can degrade the detergents containing highly branched chains.
(iii) Some synthetic detergents can give foam even in ice cold water.
(iv) Synthetic detergents are not soaps.
Explain why some times foaming is seen in river water near the place where sewage water is poured after treatment?
Draw the diagram showing micelle formation by the following detergent.
\[\ce{CH3(CH2)10CH2OS\overset{-}{O}3\overset{+}{N}a}\]
Define the following:
Cationic detergents
Explain the Following Term with Suitable Examples.
Cationic Detergents
Explain the following term with suitable example.
Cationic detergents