Advertisements
Advertisements
Question
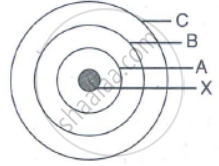
In the figure given alongside
(a) Name the shells denoted by A,B, and C. Which shell has least energy
(b) Name X and state the charge on it
(c) The above sketch is of …………. Model of an atom
Solution
(a) A is for K shell or I shell.
B is for L shell or II shell.
C is for M shell or III shell.
Shell K has the minimum amount of energy.
(b) X is a nucleus; it is positively charged.
(c) The above sketch is of Bohr model of an atom.
APPEARS IN
RELATED QUESTIONS
Compare all the proposed models of an atom given in this chapter.
Name the scientists who described the arrangement of electrons in an atom.
Name the central part of an atom where protons and neutrons are held together.
Describe Bohr's model of the atom. How did Neils Bohr explain the stability of atom?
State true or false. If false, correct the statement.
Smaller the size of the orbit, lower is the energy of the orbit.
Explain the postulates of Bohr’s atomic model.
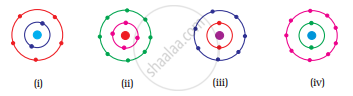
Which of the following in Fig. 4.2 do not represent Bohr’s model of an atom correctly?
What are the Limitations of Bohr’s Model?
The atomic model based on quantum theory was first proposed by ______.
Bohr's model of atoms ______.
