Advertisements
Advertisements
Question
In which of the following, functional group isomerism is not possible?
Options
Alcohols
Aldehydes
Alkyl halides
Cyanides
Solution
Alkyl halides
Explanation:
Alkyl halides do not show functional isomerism. Alcohols and ethers, aldehydes and ketones, cyanides and isocyanides are functional isomers.
APPEARS IN
RELATED QUESTIONS
What effect does branching of an alkane chain has on its boiling point?
Find out the type of isomerism exhibited by the following pair.
\[\begin{array}{cc}
\ce{CH3 - CH - CH2 - CH3 and CH3 - CH2 - O - CH2 - CH3}\\|\phantom{...........................................}\\
\ce{OH}\phantom{.........................................}\end{array}\]
Find out the type of isomerism exhibited by the following pair.
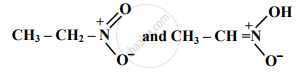
Choose the correct option.
Which type of isomerism is possible in CH3 CHCHCH3?
What type(s) of isomerism is(are) shown by [Co(NH3)4Br2]Cl?
Which of the following is a functional isomer of pentan-2-ol?
The type of isomerism possible in 2-butene is ____________.
Which of the following pairs are not functional group isomers?
| I. | \[\begin{array}{cc} \phantom{.......................}\ce{O}\\ \phantom{.......................}||\\ \ce{CH3 - CH2 - CH2 - CH2 - C - H} \end{array}\] |
| II. | \[\begin{array}{cc} \phantom{.................}\ce{O}\\ \phantom{.................}||\\ \ce{CH3 - CH2 - CH2 - C - H} \end{array}\] |
| III. | \[\begin{array}{cc} \ce{CH3 - CH2 - C - CH2 - CH3}\\ \phantom{}||\\ \phantom{}\ce{O} \end{array}\] |
| IV. | \[\begin{array}{cc} \ce{CH3 - CH - CH2 - C - H}\\ \phantom{...}|\phantom{............}||\phantom{}\\ \phantom{...}\ce{CH3}\phantom{.........}\ce{O}\phantom{} \end{array}\] |
(i) II and III
(ii) II and IV
(iii) I and IV
(iv) I and II
Compounds with same molecular formula but differing in their structures are said to be structural isomers. What type of structural isomerism is shown by
CH3 – S – CH2 – CH2 – CH3
And
\[\begin{array}{cc}
\phantom{.....................}\ce{CH3}\\
\phantom{................}/\\
\phantom{}\ce{CH3 - S - CH}\\
\phantom{...............}\backslash\\
\phantom{....................}\ce{CH3}
\end{array}\]
Assertion (A): The compound cyclooctane has the following structural formula: ![]()
It is cyclic and has conjugated 8π-electron system but it is not an aromatic compound.
Reason (R): (4n + 2)π electrons rule does not hold good and ring is not planar.
Tautomerism is exhibited by ______.
Which of the following does NOT exhibit geometrical isomerism?
Ether and alcohol are ______.
Which type of isomerism can not be shown by benzaldoxime?
Acetamide is isomer of ______.
How many structural isomers possible of the molecular formula C3H6O (excluding enol form)?
The number of acyclic structural isomers (including geometrical isomers) for pentene are ______.
Which of the following pairs of compounds is an example of position isomerism?
