Advertisements
Advertisements
Question
Match the structures of compounds given in Column I with the classes of compounds given in Column II.
| Column I | Column II | |
| (i) | \[\begin{array}{cc} \ce{CH3 - CH - CH3}\\ |\phantom{..}\\ \ce{X}\phantom{..} \end{array}\] |
(a) Aryl halide |
| (ii) | \[\ce{CH2 = CH - CH2 - X}\] | (b) Alkyl halide |
| (iii) | 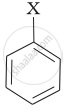 |
(c) Vinyl halide |
| (iv) | \[\ce{CH2 = CH - X}\] | (d) Allyl halide |
Solution
| Column I | Column II | |
| (i) | \[\begin{array}{cc} \ce{CH3 - CH - CH3}\\ |\phantom{..}\\ \ce{X}\phantom{..} \end{array}\] |
(b) Alkyl halide |
| (ii) | \[\ce{CH2 = CH - CH2 - X}\] | (d) Allyl halide |
| (iii) |  |
(a) Aryl halide |
| (iv) | \[\ce{CH2 = CH - X}\] | (c) Vinyl halide |
Explanation:
(i) In alkyl halide, the halogen atom is bonded to sp3 hybridised carbon atom, which may be further bonded to one, two or three alkyl groups, i.e., \[\begin{array}{cc}
\ce{CH3 - CH - CH3}\\
|\phantom{..}\\
\ce{X}\phantom{..}
\end{array}\]
(ii) Allyl halides are the compounds in which the halogen atom is bonded to sp3 hybridised carbon atom next to the carbon-carbon double bond, i.e., \[\ce{CH2 = CH - CH2 - X}\]
(iii) Aryl halides are the compounds in which the halogen atom is bonded to the sp2 hybridised carbon atom of an aromatic ring, i.e., \[\ce{C6H5X}\]
(iv) Vinyl halides are the compounds in which the halogen atom is bonded to an sp2 hybridised carbon atom of a carbon-carbon double bond, i.e., \[\ce{CH2 = CH-X}\]
APPEARS IN
RELATED QUESTIONS
Draw the structure of major monohalo products in the following reaction:
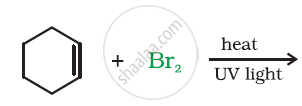
Name the following halide according to the IUPAC system and classify it as an alkyl, allyl, benzoyl (primary, secondary, tertiary), vinyl or aryl halide:
(CH3)3CCH2CH(Br)C6H5
The position of \[\ce{-Br}\] in the compound in \[\ce{CH3CH = CH(Br)(CH3)2}\] can be classified as ______.
The reaction of toluene with chlorine in the presence of iron and in the absence of light yields ______.
Name the following halide according to IUPAC system and classify it as alkyl, allyl, benzyl (primary, secondary, tertiary), vinyl or aryl halide:
\[\ce{CH3C(C2H5)2CH2Br}\]
Name the following halides according to the IUPAC system and classify them as alkyl, allyl, benzyl (primary, secondary, tertiary), vinyl or aryl halides:
CH3C(C2H5 )2CH2Br
Name the following halides according to IUPAC system and classify them as alkyl, allyl, benzyl (primary, secondary, tertiary), vinyl or aryl halides:
\[\ce{CH3CH2 C(CH3)2CH2I}\]
Name the following halide according to IUPAC system and classify them as alkyl, allyl, benzyl (primary, secondary, tertiary), vinyl or aryl halide:
\[\ce{CH3C(C2H5)2CH2Br}\]
Name the following halide according to IUPAC system and classify as alkyl, allyl, benzyl (primary, secondary, tertiary), vinyl or aryl halides:
CH3CH2C(CH3)2CH2l
Write the structure of the following organic halogen compound.
4-tert-Butyl-3-iodoheptane
