Advertisements
Advertisements
Question
Name an element which reacts with hydrogen to form a compound which is strongly acidic in water.
Solution
Hydrogen reacts with chlorine to form hydrogen chloride.
APPEARS IN
RELATED QUESTIONS
State your observation When moist starch iodide paper is introduced into chlorine gas.
Explain why dilute hydrochloric acid cannot be concentrated by boiling beyond 22.2%.
Write a fully balanced of the following case :
Magnesium metal is treated with dilute hydrochloric acid.
Give a chemical test to distinguish between the following pair of compounds:
Hydrogen chloride gas and hydrogen sulphide gas
Identify the salts P and Q from the observation given below:
When dilute HCl is added to a salt Q, a brisk effervescence is produced and the gas turns lime water milky. When NH4OH soltion is added to the above mixture (after adding dilute HCl), it produces a white precipitate which is soluble in excess NH4OH solution.
Write the balanced equation for the reaction of dilute hydrochloric acid with the following:
Sodium hydrogen carbonate
Write the balanced equation for the reaction of dilute hydrochloric acid with the following:
Magnesium sulphite
Write observation:
A small piece of zinc is added to dilute hydrochloric acid.
State your observation when:
Decomposition of bicarbonates by dil. H2SO4
2NaHCO3 + H2SO4 → Na2SO4 + 2H2O + 2CO2
2KHCO3 + H2SO4 → K2SO4 + 2H2O + 2CO2
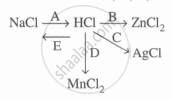
Study the flow chart and give balanced equations with conditions for the conversions A, B, C, D, and E.