Advertisements
Advertisements
Question
Phenolphthalein is a synthetic type of indicator.
Options
True
False
Solution
Phenolphthalein is a synthetic type of indicator - True
APPEARS IN
RELATED QUESTIONS
| Column A | Column B | ||
| i | eosin | 1 | losing hydrogen |
| ii | oxidation | 2 | synthetic indicator |
| 3 | losing oxygen | ||
| 4 | natural indicator |
A solution reacts with crushed egg-shells to give a gas that turns lime-water milky. The solution contains ______.
Write a word equation and then a balanced equation for the reaction taking place when:
Dilute sulphuric acid reacts with aluminium powder.
Complete and balance the followingchemicalequations:
NaHCO3 (S) + HCI (aq) →
What happens when an acid reacts with a metal?
What happens when an acid reacts with a base?
With the help of labelled diagrams, describe an activity to show that acids produce ions only in aqueous solutions.
A solution reacts with zinc granules to give a gas which burns with a 'pop' sound. The solution contains:
(a) Mg(OH)2
(b) Na2CO3
(c) NaCl
(d) HCl
On adding dilute hydrochloric acid to copper oxide powder, the solution formed is blue-green.
On the basis of the above reaction, what can you say about the nature of copper oxide?
What ions are present in the solutions of following substances? (write the symbols only)
Hydrochloric acid
What ions are present in the solutions of following substances? (write the symbols only)
Potassium hydroxide
Write the chemical formula of sodium carbonate decahydrate.
If you take some distilled water in a test-tube, add an equal amount of acetic acid to it, shake the test-tube well and leave it undisturbed on the test-tube stand, then after about 5 minutes what would you observe?
(A) There is a layer of water over the layer of acetic acid.
(B) A precipitate is setting at the bottom of the test-tube.
(C) Bubbles of colourless gas are coming out of the test-tube.
(D) There is a clear, colourless transparent solutions in the test-tube.
When _______________ is passed through fresh lime water, it turns milky.
Magnesium hydroxides are used for treating _______
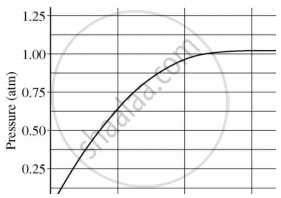
A student added 10 g of calcium carbonate in a rigid container, secured it tightly and started to heat it. After some time, an increase in pressure was observed, the pressure reading was then noted at intervals of 5 mins and plotted against time, in a graph as shown below. During which time interval did maximum decomposition take place?
A sample of soil is mixed with water and allowed to settle. The clear supernatant solution turns the pH paper yellowish-orange. Which of the following would change the colour of this pH paper to greenish-blue?
What happens when nitric acid is added to egg shell?
Acids present in fruits and vegetables are called ______ acids.
