Advertisements
Advertisements
प्रश्न
Phenolphthalein is a synthetic type of indicator.
पर्याय
True
False
उत्तर
Phenolphthalein is a synthetic type of indicator - True
APPEARS IN
संबंधित प्रश्न
When phenolphthalein is added to NaOH, the colour of the solution will become _________.
- colourless
- red
- pink
- yellow
Write a word equation and then a balanced equation for the reaction taking place when:
Dilute sulphuric acid reacts with aluminium powder.
Name one natural source of the following acids:
Lactic acid
Complete and balance the following chemical equations:
Zn (s) + HCI (aq) →
Complete and balance the following chemical equations:
NaOH (aq) HCI (aq)→
Complete and balance the following chemical equations:
CuO (s) + HCI (aq) →
What happens when an acid reacts with a base?
What ions are present in the solutions of following substances? (write the symbols only)
Hydrochloric acid
State two uses each of the following compounds:
Sodium hydroxide
Read the following statements:
I. When a red litmus paper is dipped into reaction mixture of a saponification reaction, it turns blue and the reaction is exothermic.
II. When a blue litmus paper is dipped into reaction mixture of a saponification reaction, its colour does not change and the reaction is exothermic.
III. When a red litmus paper is dipped into reaction mixture of a saponification reaction, its colour does not change and the reaction is endothermic.
IV. When a blue litmus paper is dipped into reaction mixture of a saponification reaction, its colour does not change and the reaction is endothermic.
Which of the above statements are correct:
(A) I, and II
(B) II and III
(C) III and IV
(D) I and IV
Explain the following:
Lead carbonate does not react with dilute HCl.
Answer the following question.
Identify the acid and base which form sodium hydrogen carbonate. Write the chemical equation in support of your answer. State whether this compound is acidic, basic, or neutral. Also, write its pH value.
Write any four physical properties of acids.
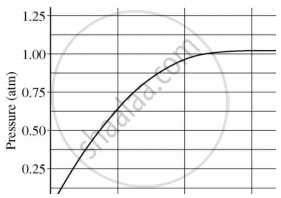
A student added 10 g of calcium carbonate in a rigid container, secured it tightly and started to heat it. After some time, an increase in pressure was observed, the pressure reading was then noted at intervals of 5 mins and plotted against time, in a graph as shown below. During which time interval did maximum decomposition take place?
During the preparation of hydrogen chloride gas on a humid day, the gas is usually passed through the guard tube containing calcium chloride. The role of calcium chloride taken in the guard tube is to ______
To protect tooth decay, we are advised to brush our teeth regularly. The nature of the tooth paste commonly used is
What are strong and weak acids? In the following list of acids, separate strong acids from weak acids.
Hydrochloric acid, citric acid, acetic acid, nitric acid, formic acid, sulphuric acid.
Solutions of acids conduct ______.
What is the correct procedure for mixing acid and water? Why?
