Advertisements
Advertisements
Question
The calorie is defined as 1 cal = 4.186 joule. Why not as 1 cal = 4 J to make the conversions easy?
Solution
One calorie is defined as the heat required to raise the temperature of 1 g of water from 14.5o C to 15.5o C. Also, exactly 4.186 J of work is to be done on heating 1 g of water from 14.5o C to 15.5o C. So, we cannot round-off 4.186 J to 4 J as it will give significant difference in work done required to raise the temperature and heat required to raise the temperature.
Since work and heat are equivalent here, so on taking 1 calorie = 4 J, the validity of the equivalence of work and heat will get defied. Thus, just to make conversion easy, 1 calorie cannot be taken equal to 4 J.
APPEARS IN
RELATED QUESTIONS
A calorimeter is kept in a wooden box to insulate it thermally from the surroundings. Why is it necessary?
Three bodies of the same material and having masses of m, m and 3m are at temperatures of 40°C, 50°C and 60°C respectively. If the bodies are brought in thermal contact, the final temperature will be:
Refer to the plot of temperature versus time (figure) showing the changes in the state of ice on heating (not to scale).
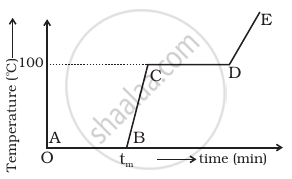
- The region AB represents ice and water in thermal equilibrium.
- At B water starts boiling.
- At C all the water gets converted into steam.
- C to D represents water and steam in equilibrium at boiling point.
On an X temperature scale, water freezes at -125°X and boils at 375°X. On a Y temperature scale, water freezes at -70°Y and boils at -30°Y. The value of temperature on X scale is ______ on which value of temperature on y Scale becomes 50° Y.
A copper block of mass 2.5 kg is heated in a furnace to a temperature of 500°C and then placed on large ice block. The maximum amount of ice that can melt is ______.
(Specific heat of copper = 0.39 Jg-1 c-1, latent heat of 0 fusion of water = 335 Jg-1)
80 gm of water at 30°C is poured on a large block of ice at 0°C. The mass of ice that melts is ______.
Work done in converting one gram of ice at -10°C into steam at 100°C is ______.
The specific heat of a substance is given by C = a + bT, where a = 1.12 kJ kg-1K-1 and b = 0.016 kJ-kg K-2. The amount of heat required to raise the temperature of 1.2 kg of the material from 280 K to 312 K is ______.
500 g of water and 100 g of ice at 0°C are in a calorimeter whose water equivalent is 40 g. 10 g of steam at 100°C is added to it. Then water in the calorimeter is ______.
(Latent heat of ice = 80 cal/g, Latent heat of steam = 540 cal/g)
540 g of ice at 0°C is mixed with 540 g of water at 80°C. The final temperature of the mixture will be ______.
