Advertisements
Advertisements
Question
The following question refers to one mole of chlorine gas.
What will happen to volume of gas, if pressure is doubled?
Solution
According to Boyle's law,
P2V2 = P1V1
or, V2 = `("P"_1"V"_1)/2`
= `("P"_1 xx 22.4)/(2"P"_1)`
= 11.2 litres
APPEARS IN
RELATED QUESTIONS
Explain Why?
Inflating a balloon seems to violate Boyle's law.
State the law which is represented by the following graph:
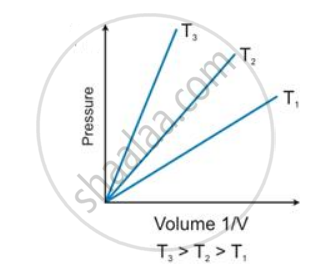
Give reasons for the following:
Inflating a balloon seems to violate Boyle's law.
Choose the correct answer:
If the pressure is doubled for a fixed mass of a gas, its volume will become
A steel cylinder of internal volume 20 litres is filled with hydrogen at 29 atmospheric pressure. If hydrogen is used to fill a balloon at 1.25 atmospheric pressure at the same temperature, what volume will the gas occupy?
Calculate the volume occupied by 2 g of hydrogen at 27°C and 4-atmosphere pressure if at STP it occupies 22.4 litres.
State Boyle's Law.
Give its
(i) mathematical expression
(ii) graphical representation and
(iii) significance.
Calculate the following:
Calculate the temperature at which a gas ‘A’ at 20°C having a volume, of 500 cc. will occupy a volume of 250 cc.
State Boyle’s law
