Advertisements
Advertisements
Question
What is the difference between the chemical composition of soaps and detergents? State in brief the action of soaps in removing an oily spot from a shirt. Why are sops not considered suitable for washing where water is hard?
Solution
Soaps are potassium or sodium salts of long chain carboxylic acid. On the other hand, detergents are ammonium or sulphonate salts of long chain carboxylic acid.
Action of soap in removing an oily spot from a shirt
The dirt present on clothes is organic in nature and insoluble in water. Therefore, it cannot be removed by only washing with water. When soap is dissolved in water, its hydrophobic ends attach themselves to the dirt and remove it from the cloth. Then, the molecules of soap arrange themselves in micelle formation and trap the dirt at the centre of the cluster. These micelles remain suspended in the water. Hence, the dust particles are easily rinsed away by water.
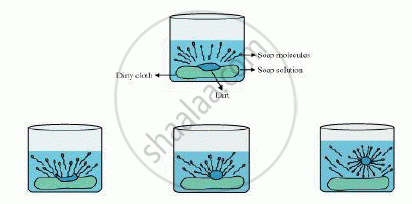
Soap does not work properly when the water is hard.
A soap is a sodium or potassium salt of long chain fatty acids. Hard water contains salts of calcium and magnesium. When soap is added to hard water, calcium and magnesium ions present in water displace sodium or potassium ions from the soap molecules forming an insoluble substance called scum. A lot of soap is wasted in the process.
RELATED QUESTIONS
A student took four test tubes P, Q, R and S and filled about 8 mL of distilled water in each. After that he dissolved an equal amount of Na2SO4 in P, K2SO4 in Q, CaSO4 in R and MgSO4 in S. On adding an equal amount of soap solution and shaking each test tube well, a good amount of lather will be obtained in the test tubes:
(a) P and Q
(b) P and R
(c) P, Q and S
(d) Q, R and S
Which of the two is better for washing clothes when the water is hard: soap or detergent? Give reason for your answer.
Give any two differences between soaps and detergents.
A student is testing water to know which is best for cleansing purposes with soaps. He would find that the cleansing action of soaps is best when he uses water obtained from
(a) rain
(b) tap
(c) hand pump
(d) pond
Explain the action of soap in removing an oily spot from a piece of cloth.
Give a scientific explanation.
Soap forms a precipitate in hard water.
Soap is a salt of ______ and sodium hydroxide.
Water, coconut oil, and _______ are necessary for soap preparation.
______ to remove strong strains on the clothes.
What are the similarities between Soap and synthetic detergent.
