Advertisements
Advertisements
Question
What do you notice when acetic acid reacts with metals?
Solution
When acetic acid reacts with metals, hydrogen is evolved (with active metals like Zn and Mg).
2CH3COOH + Zn → (CH3COO)2Zn + H2
APPEARS IN
RELATED QUESTIONS
A student puts a drop of reaction mixture of a saponification reaction first a blue litmus paper and then on a red litmus paper. He may observe that:
(a) There is no change in the blue litmus paper and the red litmus paper turns white.
(b) There is no change in the red litmus paper and the blue litmus paper turns red.
(c) There is no change in the blue litmus paper and the red litmus paper turns blue.
(d) No change in colour is observed in both the litmus papers
Name the following:
The distinctive reaction that takes place when ethanol is treated with acetic acid.
Why is the conversion of ethanol into ethanoic acid an oxidation reaction?
What is an oxidising agent?
Name two oxidising agents which can oxidise ethanol to ethanoic acid.
State any two uses of esters.
What do you observe when acetic acid is added to sodium bicarbonate?
CH3–CH2–CHO : propanal : : CH3–COOH : _______
Vinegar is a solution of ______.
Observe the diagram given below and answer the questions:
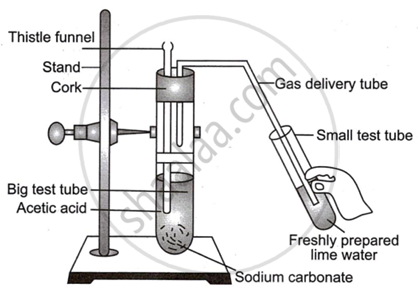
- Name the reactants in this reaction.
- Which gas comes out as effervescence in the bigger test tube?
- What is the colour change in the lime water?
- In the above experiment instead of sodium carbonate which chemical can be used to get same products?
- Write the use of acetic acid.
