Advertisements
Advertisements
Question
What is an electrochemical series? How is it useful in predicting whether a metal can liberate hydrogen from acid or not?
Solution
It is a series or table in which the various electrodes are arranged in the increasing order of standard reduction potential values. With the help of the electrochemical series, we can predict whether a given metal can liberate hydrogen from acid or not. The metals which lie above hydrogen in the electrochemical series i.e. having negative standard reduction potential values can liberate hydrogen from dilute acids whereas metals which lie below hydrogen in the electrochemical series i.e., have positive standard reduction potential values cannot liberate hydrogen from dilute acids
APPEARS IN
RELATED QUESTIONS
Fill in the blanks by choosing the appropriate word/words from those given in the brackets :
(zero, first, second, increased, decreased, anode, cathode, active, inactive, potassium cyanide, internal, external, dependent, independent, red, benzoic acid, benzoin, common ion effect, salt hydrolysis, alkali, potassium hydroxide,)
(i) In a galvanic cell, electrons flow from ………. to ……….. through the connecting wires.
(ii) Racemic mixtures are optical …………. because of …………… compensation.
(iii) The half-life period of a …………. order reaction is …………. of the concentration of the reactant.
(iv) Benzaldehyde when treated with an alcoholic solution of ……… forms ………..
(v) The solubility of calcium oxalate is …………. in the presence of ammonium oxalate because of ……… .
The following electrochemical cell is set up at 298 K:
`"Zn"//"Zn"^(2+)("aq")(1"M")////"Cu"^(2+)("aq")(1"M")//"Cu"`
Given`-> "E"_("Zn"^(2+)//"2n")^0 = -0.761"V", "E"_("Cu"^(2+)//"Cu")^0 = +0.339"V"`
Write the cell reaction.
The following electrochemical cell is set up at 298 K:
`"Zn"//"Zn"^(2+)("aq")(1"M")////"Cu"^(2+)("aq")(1"M")//"Cu"`
Given`-> "E"_("Zn"^(2+)//"2n")^0 = -0.761"V", "E"_("Cu"^(2+)//"Cu")^0 = +0.339"V"`
Calculate the emf and free energy change at 298 K.
Study the diagram given below that represents Cu-Ag electrochemical cell and answer the questions that follow.
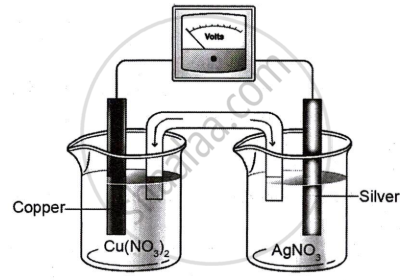
Given \[\ce{E^0_{(Cu^{2+}/Cu)}}\] = 0.337V; \[\ce{E^0_{(Ag^+/Ag)}}\] = 0.799V
- Write the cell reaction for the above cell.
- Calculate the standard emf of the cell.
- If the concentration of [Cu2+] is 0.1 M and Ecell is 0.422 V, at 25°C, calculate the concentration of [Ag+].
- Calculate ΔG for the cell.
Copper sulphate solution cannot be stored in a zinc pot. Why?
