Advertisements
Advertisements
Question
What is the action of following on phenol at low temperature?
- dil. HNO3
- conc. H2SO4
- Br2/CS2
Solution
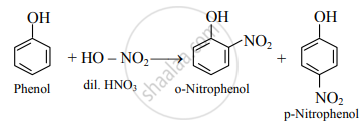
- Dilute HNO3: Phenol reacts with dilute nitric acid at low temperature to give a mixture of ortho- and para-nitrophenol.
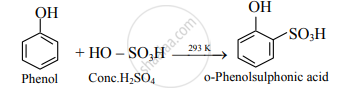
- Conc. H2SO4: At room temperature (298 K), phenol reacts with concentrated sulphuric acid to form o-phenolsulphonic acid.
- Br2 in CS2: When a reaction is carried out in a solvent of lower polarity like CS2 a mixture of ortho- and para-bromophenol is formed.

RELATED QUESTIONS
Write two points of difference between the properties of phenol and ethyl alcohol.
Give the reagents and conditions necessary to prepare phenol from Chlorobenzene.
Answer in brief.
Give the equations of the reactions for the preparation of phenol from isopropyl benezene.
The reagents used to convert phenol to 2,4,6-tribromophenol is _____________
Write IUPAC name of crotonyl alcohol.
Write the reactions involved in the preparation of phenol from benzene sulfonic acid.
An unknown alcohol is treated with Lucas reagent. Explain how you will determine whether the alcohol is primary, secondary or tertiary. Indicate by chemical equation the reaction between isopropyl alcohol and Lucas reagent.
How will you bring about the following conversions?
isopropyl alcohol to acetone
What is the action of conc. H2SO4 on carbolic acid at 373 K.
\[\ce{C3H8O ->[KMnO4][(Oxidation)] C3H6O2}\]
The compound C3H8O is a/an ____________.
Which alcohol is difficult to oxidise?
Sodium metal with ethyl alcohol gives __________ gas.
Phenol is ____________.
Which of following elements does not react with hot concentrated sulphuric acid?
+I effect of alkyl groups in alcohols increases the stability of ____________.
In Raschig's method for synthesis of phenol, the reactants used are ____________.
Identify the product B in following conversion.
\[\ce{Chlorobenzene + H2O ->[Cu, 673 K][Pressure] A ->[conc. H2SO4][373 K] B}\]
The number of σ bonds in carbolic acid is ______.
Which among the following is allylic secondary alcohol?
Identify 'Z' in the following series of reaction:
\[\ce{Butan - 2 - ol ->[PCl3] X ->[alco. KOH] Y ->[i) H2SO4][ii) H-OH/heat] Z}\]
Identify the reagent A used in the following conversion.
\[\ce{Phenol ->[A] Benzoquinone}\]
Arrange the following compounds in an increasing order of their solubility in water:
Write the product when 1°, 2° and 3° alcohol vapours are passed over hot copper.
Convert the following :
cumene to phenol.
