Advertisements
Advertisements
Questions
Write two points of difference between the properties of phenol and ethyl alcohol.
Write four points of difference between the properties of phenol and ethyl alcohol.
Solution
| Phenol | Ethyl alcohol | |
| i. | Phenol is a low melting solid. | Ethyl alcohol is liquid. |
| ii. | The aqueous solution of phenol turns blue litmus to red, i.e., phenol is weakly acidic. | The aqueous solution of ethyl alcohol is neutral to litmus, i.e, ethyl alcohol is neutral. |
| iii. | Phenol reacts with aqueous NaOH to form sodium phenoxide. | Ethyl alcohol does not react with aqueous NaOH. |
| iv. |
Phenol reacts with neutral ferric chloride solution to give deep purple colouration of ferric phenoxide. |
Ethyl alcohol does not react with neutral ferric chloride. |
RELATED QUESTIONS
Answer in one sentence/ word.
Hydroboration-oxidation of propene gives _________.
Answer in brief.
Give the equations of the reactions for the preparation of phenol from isopropyl benezene.
Reaction between Grignard reagent and aldehyde other than formaldehyde leads to formation of _______________
The reagents used to convert phenol to 2,4,6-tribromophenol is _____________
Write Kolbe’s reaction.
What is the action of following on phenol at low temperature?
- dil. HNO3
- conc. H2SO4
- Br2/CS2
Write chemical reactions to convert phenol into salicyaldehyde. Write the name of the reaction. What happens when if CCl4 is used instead of CHCl3 in the above reaction.
Phenol reacts with concentrated nitric acid in the presence of cone. H2SO4 to form ____________.
\[\ce{C3H8O ->[KMnO4][(Oxidation)] C3H6O2}\]
The compound C3H8O is a/an ____________.
____________ is used for silvering mirrors.
Bromination of phenol, will NOT give:
Phenol is ____________.
Which of following elements does not react with hot concentrated sulphuric acid?
What is the product of the following reaction?
\[\ce{CH3 - CH2 - CH2 - OH ->[conc. H2SO4][\Delta]}\]
The product 'C' in the following reaction is:
\[\ce{CH3CH2Br ->[alc. KCN] {'A'} ->[H3O^+][\Delta] {'B'} ->[i. LiAlH4][ii. H3O^+] {'C'}}\]
+I effect of alkyl groups in alcohols increases the stability of ____________.
Phenoxide ion is more stable than phenol due to the ____________.
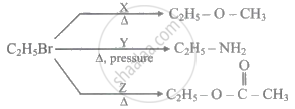
Identify reagents X, Y and Z.
Which of the following alcohols has tertiary allylic carbon?
Which of the following reagents is used to convert phenol to benzene?
Identify the product B in following conversion.
\[\ce{Chlorobenzene + H2O ->[Cu, 673 K][Pressure] A ->[conc. H2SO4][373 K] B}\]
Which among the following is allylic secondary alcohol?
Identify the alcohol that reacts immediately with Lucas reagent?
Identify the compound having highest boiling point from the following?
The compound which reacts fastest with Lucas reagent at room temperature is ______.
The acid, which contains both -OH and -COOR groups is ______.
Write the chemical reaction when hot copper is treated with Vapours of 1° (primary) alcohol.
Convert the following :
cumene to phenol.
