Advertisements
Advertisements
Question
Write chemical reactions to convert phenol into salicyaldehyde. Write the name of the reaction. What happens when if CCl4 is used instead of CHCl3 in the above reaction.
Solution
- Phenol to salicyaldehyde: When phenol is treated with chloroform in an aqueous sodium hydroxide solution followed by hydrolysis with acid, salicylaldehyde is formed.
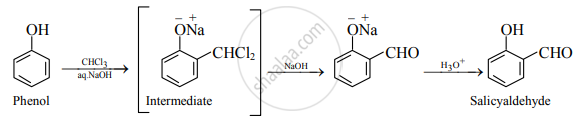
- This reaction is known as the Reimer-Tiemann reaction.
- If the solvent chloroform (CHCl3) is replaced by carbon tetrachloride (CCl4), salicylic acid is formed.
APPEARS IN
RELATED QUESTIONS
Write two points of difference between the properties of phenol and ethyl alcohol.
Answer in brief.
Explain why p-nitrophenol is a stronger acid than phenol.
Answer in brief.
Give the equations of the reactions for the preparation of phenol from isopropyl benezene.
Oxidation of ethyl alcohol using K2Cr2O7/dil H2SO4 leads to formation of _______________
When vapours of tert.butyl alcohol are passed over hot copper, it gives _____________
Reaction between hot conc. HI and anisole gives ______________
Write the name of major product when anisole reacts HI at 398 K
Write Kolbe’s reaction.
Write chemical equation of acetyl chloride with phenol
Write the reactions for the preparation of carbolic acid from aniline.
An organic compound gives hydrogen on reaction with sodium metal. It forms an aldehyde with molecular formula C2H4O on oxidation with pyridinium chlorochromate. Give the chemical equations in support of these observations.
\[\ce{C3H8O ->[KMnO4][(Oxidation)] C3H6O2}\]
The compound C3H8O is a/an ____________.
Sodium metal with ethyl alcohol gives __________ gas.
Bromination of phenol, will NOT give:
Carbolic acid is oxidised by acidified sodium dichromate to give ______.
In Dow process, chlorobenzene is converted into phenol by ____________.
____________ will NOT undergo acetylation.
Which of the following reagents is used to convert phenol to benzene?
Which among the following is allylic secondary alcohol?
Which of the following compounds does NOT react with sodium metal?
Which of the following on oxidation yields ethyl methyl ketone?
Identify product B in the following conversion?
\[\ce{Phenol ->[NaOH] A ->[CH3I] B}\]
The product C in the following reaction is

Arrange O - H, C - H and N - H bonds in increasing order of their bond polarity.
Arrange the following compounds in an increasing order of their solubility in water:
Explain the reaction of 1° and 2° alcohol with oxidising agent chromic anhydride (CrO3).
Write the chemical reaction when hot copper is treated with Vapours of 2° (secondary) alcohol.
Convert the following :
cumene to phenol.
