Advertisements
Advertisements
Question
Which of the following is a wrong description of binding energy of a nucleus?
Options
It is the energy required to break a nucleus into its constituent nucleons.
It is the energy made available when free nucleons combine to form a nucleus.
It is the sum of the rest mass energies of its nucleons minus the rest mass energy of the nucleus.
It is the sum of the kinetic energy of all the nucleons in the nucleus.
Solution
It is the sum of the kinetic energies of all the nucleons present in the nucleus.
Binding energy of a nucleus is defined as the energy required to break the nucleus into its constituents. It is also measured as the Q-value of the breaking of nucleus, i.e. the difference between the rest energies of reactants (nucleus) and the products (nucleons) or the difference between the kinetic energies of the products and the reactants.
APPEARS IN
RELATED QUESTIONS
In a typical nuclear reaction, e.g.
`"_1^2H+"_1^2H ->"_2^3He + n + 3.27 \text { MeV },`
although number of nucleons is conserved, yet energy is released. How? Explain.
Write the relationship between the size of a nucleus and its mass number (A)?
Using the curve for the binding energy per nucleon as a function of mass number A, state clearly how the release in energy in the processes of nuclear fission and nuclear fusion can be explained.
Suppose we have 12 protons and 12 neutrons. We can assemble them to form either a 24Mg nucleus or two 12C nuclei. In which of the two cases more energy will be liberated?
The mass number of a nucleus is equal to
As the mass number A increases, the binding energy per nucleon in a nucleus
In one average-life,
For nuclei with A > 100,
(a) the binding energy of the nucleus decreases on an average as A increases
(b) the binding energy per nucleon decreases on an average as A increases
(c) if the nucleus breaks into two roughly equal parts, energy is released
(d) if two nuclei fuse to form a bigger nucleus, energy is released.
Assume that the mass of a nucleus is approximately given by M = Amp where A is the mass number. Estimate the density of matter in kgm−3 inside a nucleus. What is the specific gravity of nuclear matter?
A neutron star has a density equal to that of the nuclear matter. Assuming the star to be spherical, find the radius of a neutron star whose mass is 4.0 × 1030 kg (twice the mass of the sun).
Calculate the mass of an α-particle. Its Its binding energy is 28.2 MeV.
(Use Mass of proton mp = 1.007276 u, Mass of `""_1^1"H"` atom = 1.007825 u, Mass of neutron mn = 1.008665 u, Mass of electron = 0.0005486 u ≈ 511 keV/c2,1 u = 931 MeV/c2.)
(a) Calculate the energy released if 238U emits an α-particle. (b) Calculate the energy to be supplied to 238U it two protons and two neutrons are to be emitted one by one. The atomic masses of 238U, 234Th and 4He are 238.0508 u, 234.04363 u and 4.00260 u respectively.
(Use Mass of proton mp = 1.007276 u, Mass of `""_1^1"H"` atom = 1.007825 u, Mass of neutron mn = 1.008665 u, Mass of electron = 0.0005486 u ≈ 511 keV/c2,1 u = 931 MeV/c2.)
What is the unit of mass when measured on the atomic scale?
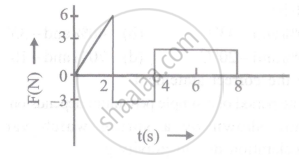
The force 'F' acting on a particle of mass 'm' is indicated by the force-time graph shown below. The change in momentum of the particle over the time interval from zero to 8s is:
A nucleus of mass M emits a γ-ray photon of frequency 'v'. The loss of internal energy by the nucleus is ______.
