Advertisements
Advertisements
Question
Which of the following does not belong to the same homologous series?
Options
CH4
C2 H6
C3 H8
C4 H8
Advertisements
Solution
C4 H8
Explanation -
The succeeding members of the homologues series differ by a —CH2 - unit. The general formula for the alkane is CnH2n+2. Methane (CH4), ethane (C2H6) and propane (C3H8) are members of homologues series for alkanes while butene (C4H8) is the fourth member of homologues series for alkene.
APPEARS IN
RELATED QUESTIONS
Write the next homologue of the following: C4H6
Classify the following carbon compounds into two homologous series and name them.
C3H4, C3H6, C4H6, C4H8, C5H8, C5H10
Write the name and formula of the 2nd member of homologous series having general formula CnH2n + 2.
Write the molecular formula of two consecutive members of homologous series of aldehydes. State which part of these compounds determines their
- physical and
- chemical properties
Write the molecular formula of first two members of homologous series having functional group – OH
Write the molecular formula of the 2nd and 3rd member of the homologous series where the first member is ethyne.
Complete the following table which relates to the homologous series of hydrocarbons.
|
General Formula |
IUPAC name of the homologous series | Characteristic bond type | IUPAC name of the first member of the series |
| `C_nH_(2n-2)` | (A)________ | (B)______ | (C)______ |
| C_nH_(2n+1) | (B)________ | (E)______ | (F)______ |
Give the structure of the second member of the alcohol group.
What is homologous series ?
What is the difference in the molecular formula of any two adjacent homologues in terms of molecular mass?
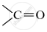 .
.
Haloalkanes react with alkalies to produce alcohol. Give the equation for the preparation of the second member of the homologous series of alcohol. State under what condition the reaction occurs.
Cyclohexane : Cyclic hydrocarbon : : Isobutylene : _______
Complete the following table for homologous series of alcohols.
| Name | Molecular formula | Condensed structural formula | Number of carbon atom | Number of -CH2- units | Boiling point °C |
| Methanol | CH4O | CH3-OH | 1 | 1 | 63 |
| Ethanol | C2H6O | CH3–CH2-OH | 2 | 2 | 78 |
| Propanol | C3H8O | CH3–CH2–CH2-OH | ______ | ______ | 97 |
| Butanol | C4H10O | CH3–CH2–CH2–CH2–OH | ______ | ______ | 118 |
Successive members of a homologous series vary by how many atomic mass unit?
A carbon compound ‘A’ having melting point 156K and boiling point 351K, with molecular formula C2H6O is soluble in water in all proportions.
- Identify ‘A’ and draw its electron dot structure.
- Give the molecular formulae of any two homologues of ‘A’.
Write the chemical formula of two consecutive homologous of organic compounds having functional group - OH.
What happens to the (i) boiling point and (ii) solubility of organic compounds of a homologous series as the molecular mass increases?
Consider the following molecular formulae of carbon compounds:
(i) CH3COOH (ii) CH3OH (iii) C2H6 (iv) C3H4 (v) C4H8
- Which one of these compounds belongs to homologous series of alcohols?
- Identify the compound having triple bond between carbon-carbon atoms.
- Write the molecular formula of the first member of the homologous series to which CH3COOH belongs.
- Write the general formula of the series to which the compound C4H8 belongs.
Name the third homologue of aldehydes.
