Advertisements
Advertisements
Question
Which of the following is not a characteristic of a crystalline solid?
Options
Definite and characteristic heat of fusion.
Isotropic nature.
A regular periodically repeated pattern of arrangement of constituent particles in the entire crystal.
A true solid.
A regular arrangement of constituent particles.
Sharp melting point.
Solution
Isotropic nature.
Explanation:
Crystalline solids are anisotropic in nature, that is some of their physical properties like electrical resistance or refractive index show different values when measured along different directions in the same crystal. This arises from the different arrangement of particles in different directions arrangement of particles along different directions
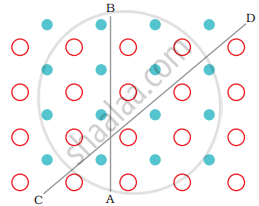
Anisotropy in crystals is due to different arrangements of particles along different directions.
Isotropy: In the case of amorphous substances, properties such as electrical conductivity, refractive index, thermal expansion, etc. are identical in all directions, just as in the case of gases or liquids. This property is called isotropy, and the substances showing this property are called isotropic.
RELATED QUESTIONS
Classify the following solids in different categories based on the nature of intermolecular forces operating in them:
Potassium sulphate, tin, benzene, urea, ammonia, water, zinc sulphide, graphite, rubidium, argon, silicon carbide.
Classify each of the following solids as ionic, metallic, molecular, network (covalent) or amorphous.
(i) Tetra phosphorus decoxide (P4O10)
(ii) Ammonium phosphate (NH4)3PO4
(iii) SiC
(iv) I2
(v) P4
(vii) Graphite
(viii) Brass
(ix) Rb
(x) LiBr
(xi) Si
Based on the nature of intermolecular forces, classify the following solids:
Sodium sulphate, Hydrogen
The existence of a substance in more than one solid modifications is known as ____________.
Which of the following is not a characteristic property of solids?
Which of the following conditions favours the existence of a substance in the solid state?
Match the items given in Column I with the items given in Column II.
| Column I | Column II |
| (i) Mg in solid state | (a) p-Type semiconductor |
| (ii) MgCl2 in molten state | (b) n-Type semiconductor |
| (iii) Silicon with phosphorus | (c) Electrolytic conductors |
| (iv) Germanium with boron | (d) Electronic conductors |
Which among the following will show anisotropy?
