Advertisements
Advertisements
Question
Which of the following statements are correct?
(i) S – S bond is present in
(ii) In peroxosulphuric acid
(iii) Iron powder along with
(iv) Change in enthalpy is positive for the preparation of
Solution
(i) S – S bond is present in
(ii) In peroxosulphuric acid
Explanation:
In
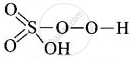
(O in peroxide linkage has oxidation state – 1)
APPEARS IN
RELATED QUESTIONS
What is molecular formula of oleum?
(a) H2SO3
(b) H2SO4
(c) H2S2O7
(d) H2S2O8
Draw the structure of the following:
H2SO4
Why is
What happens when dilute sulphuric acid is treated with CaF2
When concentrated sulphuric acid was added to an unknown salt present in a test tube a brown gas (A) was evolved. This gas intensified when copper turnings were added to this test tube. On cooling, the gas (A) changed into a colourless solid (B).
1) Identify (A) and (B).
2) Write the structures of (A) and (B).
3) Why does gas (A) change to solid on cooling?
The molecular formula of oleum is ____________.
Which among the following catalysts is used in manufacture of sulphuric acid by contact process?
What is the ratio of volumes of gases involved in the preparation of sulphur trioxide from sulphur dioxide and dioxygen respectively under similar conditions of temperature and pressure?
The number of dative bonds in sulphric acid molecule is ______.
Write the reaction of conc. H2SO4 with sugar. What is the role of H2SO4 in this reaction?
