Advertisements
Advertisements
Question
Why are carboxylic acids more acidic than alcohols or phenols although all of them have hydrogen atom attached to a oxygen atom \[\ce{(-O-H)}\]?
Solution
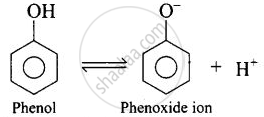
The aliphatic carboxylic acids are stronger acids than alcohols and phenols. The difference in the relative acidic strengths can be understood if we compare the resonance hybrids of carboxylate ion and ethoxide phenoxide ion.
\[\ce{RCOOH ⇌ RCOO- + H+}\]
\[\ce{ROH ⇌ RO- + H+}\]
The resonance hybrids may be represented as:
\[\begin{array}{cc}
\phantom{.............}\ce{O^{δ-}}\\
\phantom{.........}//\\
\ce{R - C}\\
\phantom{........}\backslash\backslash\\
\phantom{............}\ce{O^{δ-}}\\
\end{array}\]
Carboxylate ion
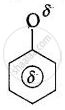
Phenoxide ion
\[\ce{RO-}\]
(No resonance)
The resonance hybrids may be represented as: Carboxylate ion Phenoxide ion (No resonance)
The electron charge on the carboxylate ion is more dispersed in comparison to the phenate ion since there are two electronegative oxygen atoms in carboxylate ion as compared to only one oxygen atom in phenoxide ion. In other words, the carboxylate ion is relatively more stable as compared to phenate ion. Thus, the release of H+ ion from carboxylic acid is comparatively easier or it behaves as a stronger acid than phenol.
APPEARS IN
RELATED QUESTIONS
Give reasons Acetylation of aniline reduces its activation effect.
Why is alpha (α) hydrogen of carbonyl compounds acidic in nature?
Which of the following compounds do not undergo aldol condensation?
(i) \[\ce{CH3 - CHO}\]
(ii) 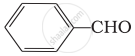
(iii) \[\begin{array}{cc}
\phantom{}\ce{O}\\
\phantom{}||\\
\ce{CH3 - C - CH3}
\end{array}\]
(iv) \[\begin{array}{cc}
\phantom{}\ce{CH3}\\
|\phantom{...}\\
\ce{CH3 - C - CHO}\phantom{..}\\
|\phantom{...}\\
\phantom{}\ce{CH3}\\
\end{array}\]
Why is there a large difference in the boiling points of butanal and butan-1-ol?
What product will be formed on reaction of propanal with 2-methylpropanal in the presence of \[\ce{NaOH}\]? What products will be formed? Write the name of the reaction also.
Assertion: The α-hydrogen atom in carbonyl compounds is less acidic.
Reason: The anion formed after the loss of α-hydrogen atom is resonance stabilised.
Give reasons to support the answer:
Presence of Alpha hydrogen in aldehydes and ketones is essential for aldol condensation.
Cross aldol condensation occurs between
Which of the following does not give aldol condensation reaction?
What is aldol condensation? Explain it with suitable examples.
