Advertisements
Advertisements
Question
Why do Free electrons not leave the metal surface on their own
Solution
Free electrons cannot leave the metal surface on their own because they do not have sufficient kinetic energy.
APPEARS IN
RELATED QUESTIONS
In a cathode ray tube state:
(i) the purpose of covering cathode by thorium and carbon.
(ii) the purpose of the fluorescent screen.
(iii) how is it possible to increase the rate of emission of electrons.
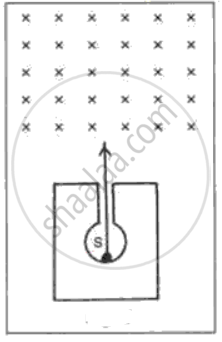
In following fig. shows a mixed source S of alpha and beta particles in a thick lead container. The particles pass through a magnetic field in a direction perpendicular to the plane of paper (inwards as shown by x). State and show in the diagram how the particles get affected.
A nucleus is \[\ce{^24_11 Na}\] β-radioactive.
Write the equation represent β-decay.
What are the types of emission?
Name a common device where a hot cathode ray tube is used.
State three common properties of beta rays and cathode rays.
State three ways of obtaining an electron-beam.
The half-life of a radioactive nuclide is 100 hours. The fraction of original activity that will remain after 150 hours would be ______.
Two radioactive sources A and B of half lives of 1 hour and 2 hours, respectively, initially contain the same number of radioactive atoms. At the end of two hours, their rates of disintegration are in the ratio of ______.
