Advertisements
Advertisements
Question
Why is benzene extra ordinarily stable though it contains three double bonds?
Solution
Benzene is a hybrid of resonating structures given as:
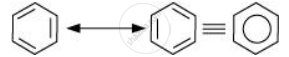
All six carbon atoms in benzene are sp2 hybridized. The two sp2 hybrid orbitals of each carbon atom overlap with the sp2 hybrid orbitals of adjacent carbon atoms to form six sigma bonds in the hexagonal plane. The remaining sp2 hybrid orbital on each carbon atom overlaps with the s-orbital of hydrogen to form six sigma C–H bonds. The remaining unhybridized p-orbital of carbon atoms has the possibility of forming three π bonds by the lateral overlap of \[\ce{C_1 - C_2, C_3 - C_4, C_5 - C_6 or C_2 - C_3, C_4 - C_5, C_6 - C_1}\]
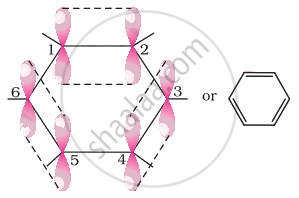
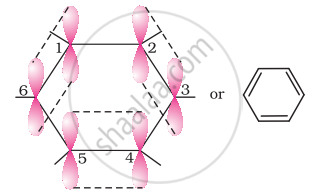
The six π’s are delocalized and can move freely about the six-carbon nuclei. Even after the presence of three double bonds, these delocalized π-electrons stabilize benzene.
APPEARS IN
RELATED QUESTIONS
Write down the products of ozonolysis of 1, 2-dimethylbenzene (o-xylene). How does the result support Kekulé structure for benzene?
Arrange the following carbanions in order of their decreasing stability.
(A) H3C – C ≡ C–
(B) H – C ≡ C–
(C) \[\ce{H3C - C\overset{-}{H2}}\]
Which of the following are correct?
(i) \[\ce{CH3 - O - CH^{⊕}2}\] is more stable than \[\ce{CH3 - CH^{⊕}2}\]
(ii) (CH3)2CH⊕ is less stable than \[\ce{CH3 - CH2 - CH^{⊕}2}\]
(iii) \[\ce{CH3 = CH2 - CH^{⊕}2}\] is more stable than \[\ce{CH3 - CH2 - CH^{⊕}2}\]
(iv) \[\ce{CH2 - CH^{⊕}}\] is more stable than \[\ce{CH3 - CH^{⊕}2}\]
How will you convert benzene into p – nitrobromobenzene
How will you convert benzene into m – nitrobromobenzene
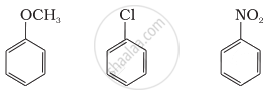
Arrange the following set of compounds in the order of their decreasing relative reactivity with an electrophile. Give reason.
The relative reactivity of 1°, 2°, 3° hydrogen’s towards chlorination is 1 : 3.8 : 5. Calculate the percentages of all monochlorinated products obtained from 2-methylbutane.
The number ratio of σ and π bonds in benzene is ______.
Mark the incorrect statement from the following:
