Advertisements
Advertisements
Question
Write a reaction to distinguish acetaldehyde from acetone
Solution 1
Tollens’ reagent test: Acetaldehyde being an aldehyde reduces Tollens’ reagent to shining silver mirror, whereas propanone being acetone does not.
\[\ce{\underset{\text{Acetaldehyde}}{CH3CHO} + \underset{\text{Tollens’ reagent}}{2[Ag(NH3)2]+} + 3OH- -> \underset{\text{Acetate ion}}{CH3COO-} + \underset{\text{Silver metal}}{2Ag ↓} + 4NH3 + 2H2O}\]
\[\ce{\underset{\text{Acetone}}{CH3COCH3} ->[Tollens 'reagent] No silver mirror}\]
Solution 2
Fehling’s solution test: Acetaldehyde reduces Fehling’s solution to red precipitate of Cu2O whereas acetone being a ketone does not.
\[\ce{\underset{\text{Acetaldehyde}}{CH3CHO} + \underset{\text{From Fehling’s solution}}{2Cu^2+ + 5OH-} ->\underset{\text{Acetate ion}}{CH3COO-} + \underset{\text{Cuprous oxide (Red ppt.)}}{Cu2O ↓} + 3H2O}\]
APPEARS IN
RELATED QUESTIONS
Write a note on Stephen reaction.
Identify the INCORRECT reaction.
If acetaldehyde is treated with Fehling's solution, the change that occurs in the system is ____________.
An aldehyde or ketone reacts with hydrogen cyanide to form corresponding cyanohydrin derivatives. This reaction is an example of ____________.
Compounds of general formula, are called ____________.
\[\begin{array}{cc} \phantom{...}\ce{R}\phantom{....}\ce{OR'}\phantom{}\\ \phantom{}\backslash\phantom{..}/\\ \ce{C}\\ \phantom{}/\phantom{..}\backslash\\ \phantom{...}\ce{H}\phantom{....}\ce{OR'}\phantom{} \end{array}\]
The compound which does NOT undergo haloform reaction is ____________.
Following reaction is an example of ____________.
\[\ce{Benzaldehyde ->[Nitration] {m}-Nitrobenzaldehyde}\]
\[\ce{Benzoic acid ->[i) NH3][ii) \Delta] A ->[NaOBr] B ->[NaNO2/HCl] C}\] ‘C’ is:
Identify the product formed in the reaction.
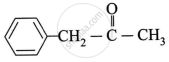
Identify A, B and C.
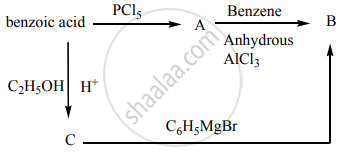
How will you convert benzaldehyde into the following compound?
Benzophenone
What is the action of HCN on ethanal?
Write the structure of the major product of the aldol condensation of benzaldehyde with acetone.
How is the following conversion effected Hex-3-yne into hexan-3-one?
How will you prepare ethyl acetate from methyl acetate?
How will you prepare lactic acid from ethanol?
How will you prepare acetaldehyde from ethyne?
Which one of the following substituents at para-position is most effective in stabilizing the phenoxide 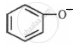 ion?
ion?
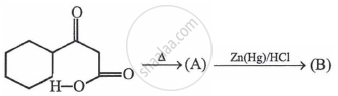
In the above reaction, product (B) is:
What is the number of different products formed when mixture of ethanal and propanal reacts with aq. NaOH after warming?
Aldehydes are readily oxidised to yield carboxylic acids but ketones are inert to oxidation. Which is the most likely explanation regarding this difference in reactivity?
A carbonyl compound A having molecular formula C5H10O forms crystalline precipitate with sodium bisulphate and gives positive iodoform test. A does not reduce Fehling solution. Identify A.
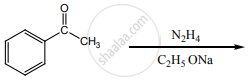
Write a note on Clemmensen reduction.
Write the structure of the products obtained from the following ketones by action of hydrazine in presence of a slightly acidic medium.
Write the structure of the major product of the aldol condensation of benzaldehyde with acetone.
Write the structure of the major product of the aldol condensation of benzaldehyde with acetone.